 Back
BackRespiration and Metabolism: Biochemical Pathways in Human Physiology
Study Guide - Smart Notes

Respiration and Metabolism
Introduction to Metabolism
Metabolism encompasses all chemical reactions occurring within the body, divided into two main categories: anabolism and catabolism. Anabolism requires energy input to synthesize large molecules, while catabolism releases energy by breaking down large molecules into smaller ones. Catabolic reactions drive the anabolic production of ATP, involving numerous oxidation-reduction reactions. Complete catabolism of glucose requires oxygen as the final electron acceptor, a process known as aerobic cellular respiration.
Glycolysis and the Lactic Acid Pathway
Glycolysis: The First Step in Glucose Catabolism
Glycolysis is the initial step in the breakdown of glucose, occurring in the cytoplasm and functioning anaerobically. The process begins with the phosphorylation of glucose, preventing its diffusion out of the cell. Glucose is then split into two molecules of pyruvic acid, with the overall reaction:
Glucose (C6H12O6) → 2 Pyruvic Acid (C3H4O3)
Net gain: 2 ATP and 2 NADH
Enzymes involved: Kinases, phosphatases, isomerases, dehydrogenases
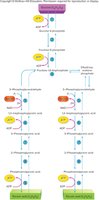
Glycolysis is exergonic, transferring energy to ADP to form ATP. The fate of pyruvic acid depends on the presence of oxygen.
Decision Point: Aerobic vs. Anaerobic Pathways
Pyruvic acid produced by glycolysis can follow one of two pathways:
Aerobic pathway: If oxygen is present, pyruvic acid enters the mitochondria for further oxidation.
Anaerobic pathway: In the absence of oxygen, pyruvic acid is converted to lactic acid.
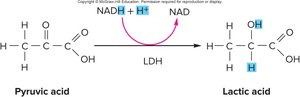
Lactic Acid Pathway (Anaerobic Metabolism)
When oxygen is unavailable, NADH donates electrons to pyruvic acid, regenerating NAD and forming lactic acid. This process, called fermentation, does not produce additional ATP beyond the 2 from glycolysis. Muscle cells can use lactic acid fermentation, but excessive lactic acid can lead to acidity and is not favored by the body. Red blood cells rely solely on this pathway due to the absence of mitochondria. Excess lactic acid is transported to the liver, where it is converted back to pyruvic acid and NADH by lactic acid dehydrogenase. Pyruvic acid can then be used to form glucose or glycogen.
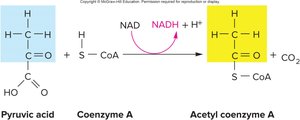
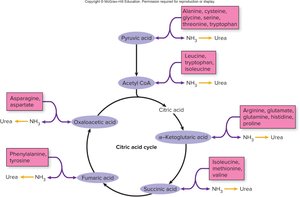
Aerobic Respiration: Citric Acid Cycle (TCA/Krebs Cycle)
Transition Step and Entry into the Citric Acid Cycle
If oxygen is present, pyruvic acid enters the mitochondrial matrix and undergoes a transition step:
Carbon dioxide is removed, forming acetic acid.
Acetic acid combines with coenzyme A to form acetyl CoA.
For each glucose: 2 acetyl CoA + 2 CO2 are produced.
Citric Acid Cycle (Krebs/TCA Cycle)
Acetyl CoA enters the citric acid cycle by combining with oxaloacetic acid to form citric acid. The cycle regenerates oxaloacetic acid and produces:
ATP via substrate-level phosphorylation
NADH and FADH2 (coenzymes capturing energy)
CO2 as a waste product
Heat (energy loss)
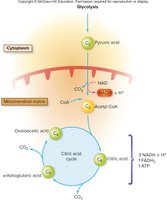
The two main coenzymes are NAD and FAD, which function as electron carriers.
Aerobic Respiration: Electron Transport Chain (ETC) and Oxidative Phosphorylation
Electron Transport Chain (ETC)
The ETC is located on the inner mitochondrial membrane and converts coenzyme energy into ATP. NADH and FADH2 donate electrons to the chain, which are passed along a series of transporters. This process pumps H+ ions into the intermembrane space, creating a concentration gradient. H+ flows back into the matrix through ATP synthase, driving ATP production.
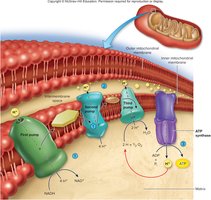
Role of Oxygen and ATP Yield
Oxygen serves as the final electron acceptor, forming water:
Theoretical ATP yield per glucose: 36–38 ATP
Actual yield: 30–32 ATP (due to energy costs of ATP export)
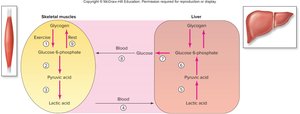
Interconversion of Glucose, Lactic Acid, and Glycogen
Glucose Storage and Glycogenesis
Cells cannot store glucose directly due to osmotic effects. Instead, glucose is phosphorylated to glucose-6-phosphate, trapping it inside the cell. Glycogenesis is the process of converting glucose-6-phosphate into glycogen for storage, primarily in the liver, skeletal muscle, and cardiac muscle.
Glycogenolysis and Glucose Release
When glucose is needed, glycogen is broken down via glycogenolysis to glucose-1-phosphate, then to glucose-6-phosphate. Only the liver can release free glucose into the bloodstream due to the enzyme glucose-6-phosphatase.
Cori Cycle and Gluconeogenesis
Excess lactic acid from muscles is transported to the liver, where it is converted to pyruvic acid and then to glucose-6-phosphate. This process, called gluconeogenesis, allows the liver to produce new glucose, which can be sent back to muscles, completing the Cori cycle.
Metabolism of Lipids and Proteins
Lipid Metabolism: Lipogenesis and Lipolysis
Excess glucose is converted to fatty acids and triglycerides via lipogenesis. When energy is needed, triglycerides are broken down into fatty acids and glycerol by lipolysis. Fatty acids undergo β-oxidation to form acetyl CoA, which enters the citric acid cycle. Glycerol can be converted to glucose via gluconeogenesis.
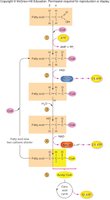
Ketogenesis
When fatty acid breakdown exceeds utilization, the liver converts acetyl CoA into ketone bodies (ketogenesis). These water-soluble molecules can accumulate, causing ketosis.
Amino Acid Metabolism
Excess amino acids can be used for energy or converted to carbohydrates or fats. Gluconeogenesis from amino acids provides glucose. If amino acids are needed, pyruvic acid and citric acid cycle intermediates can be converted to amino acids by transamination.
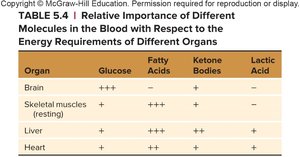
Summary: Energy Source Preferences by Organ
Relative Importance of Energy Molecules
Different organs prefer different energy sources. The brain relies heavily on glucose, while muscles and the liver can utilize fatty acids, ketone bodies, and lactic acid.
Organ | Glucose | Fatty Acids | Ketone Bodies | Lactic Acid |
|---|---|---|---|---|
Brain | +++ | – | + | – |
Skeletal muscles (resting) | + | +++ | + | – |
Liver | ++ | ++ | + | + |
Heart | + | ++ | ++ | + |
Additional info: These notes expand on the original content with definitions, examples, and academic context to ensure completeness and clarity for biochemistry students.
