Back
BackRespiration and Metabolism: Biochemical Pathways of Energy Production
Study Guide - Smart Notes

Respiration and Metabolism
Overview of Metabolism
Metabolism encompasses all chemical reactions occurring within the body, divided into two main categories: anabolism (energy-requiring synthesis of large molecules) and catabolism (energy-releasing breakdown of large molecules). Catabolic reactions, such as the breakdown of glucose, fatty acids, and amino acids, provide the energy necessary for ATP production, which in turn powers anabolic processes. These reactions involve numerous oxidation-reduction steps, with aerobic cellular respiration requiring oxygen as the final electron acceptor.
Glycolysis and the Lactic Acid Pathway
Glycolysis: The First Step in Glucose Catabolism
Glycolysis is the initial metabolic pathway for glucose catabolism, occurring in the cytoplasm and not requiring oxygen (anaerobic). It involves the splitting of a 6-carbon glucose molecule into two 3-carbon pyruvic acid molecules. The process requires the input of 2 ATP to phosphorylate glucose, preventing its diffusion out of the cell. Glycolysis results in a net gain of 2 ATP, 2 NADH, and 2 pyruvic acid molecules per glucose molecule.
Key Enzymes: Kinases (phosphate transfer), isomerases (rearrangement), dehydrogenases (hydrogen removal).
Net Reaction:
Location: Cytoplasm
Products: 2 pyruvic acid, 2 NADH, 2 ATP (net)
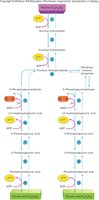
Fate of Pyruvic Acid: Aerobic vs. Anaerobic Pathways
The pathway chosen by pyruvic acid depends on the presence or absence of oxygen. In the presence of oxygen, pyruvic acid enters the mitochondria for aerobic respiration. In the absence of oxygen, it undergoes anaerobic fermentation to form lactic acid.
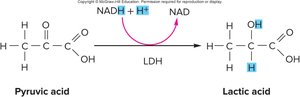
Lactic Acid Pathway (Anaerobic Metabolism/Fermentation)
When oxygen is unavailable, NADH donates its electrons to pyruvic acid, regenerating NAD+ and converting pyruvic acid to lactic acid. This process, catalyzed by lactate dehydrogenase, does not produce additional ATP beyond the 2 ATP from glycolysis. Lactic acid can accumulate, especially in muscle cells, and is transported to the liver for conversion back to pyruvic acid or glucose (Cori cycle).
Equation:
Significance: Allows glycolysis to continue in the absence of oxygen by regenerating NAD+.
Limitations: Low ATP yield; lactic acid buildup can cause acidosis.
Special Cases: Red blood cells rely solely on lactic acid fermentation due to lack of mitochondria.
Aerobic Respiration: Citric Acid Cycle (TCA/Krebs Cycle)
Transition Step and Entry into the Citric Acid Cycle
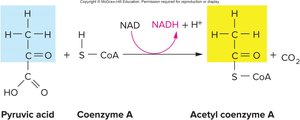
In the presence of oxygen, pyruvic acid is transported into the mitochondrial matrix, where it undergoes decarboxylation to form acetic acid. Acetic acid combines with coenzyme A to form acetyl CoA, which enters the citric acid cycle.
Transition Reaction:
Location: Mitochondrial matrix
Purpose: Links glycolysis to the citric acid cycle by producing acetyl CoA
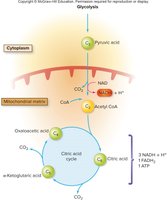
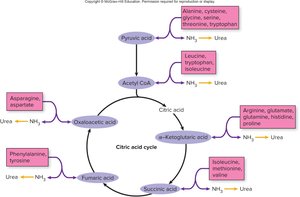
Citric Acid Cycle (Krebs/TCA Cycle)
The citric acid cycle is a series of enzyme-catalyzed reactions that oxidize acetyl CoA to CO2 and transfer high-energy electrons to NAD+ and FAD, forming NADH and FADH2. It also produces a small amount of ATP via substrate-level phosphorylation.
Key Steps: Acetyl CoA combines with oxaloacetic acid to form citric acid, which is then metabolized back to oxaloacetic acid, releasing CO2 and capturing energy in NADH, FADH2, and ATP.
Products per glucose: 6 NADH, 2 FADH2, 2 ATP, 4 CO2
Coenzymes: NAD+ and FAD (function as electron carriers)
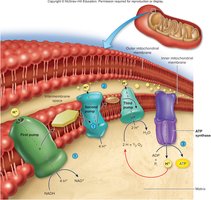
Electron Transport Chain and Oxidative Phosphorylation
Electron Transport Chain (ETC)
The ETC is located on the inner mitochondrial membrane and is responsible for converting the energy stored in NADH and FADH2 into ATP. Electrons are passed through a series of protein complexes, pumping protons into the intermembrane space and creating a proton gradient. ATP synthase uses this gradient to synthesize ATP from ADP and inorganic phosphate.
Final Electron Acceptor: Oxygen, which combines with electrons and protons to form water.
ATP Yield: Theoretically, each NADH yields 2.5–3 ATP, and each FADH2 yields 1.5–2 ATP.
Overall Reaction:
ATP Balance Sheet
Substrate-level phosphorylation: 4 ATP (2 from glycolysis, 2 from citric acid cycle)
Oxidative phosphorylation: 26–34 ATP (varies by cell type and conditions)
Total theoretical yield: 36–38 ATP per glucose
Actual yield: 30–32 ATP per glucose (due to energy costs of transporting ATP out of mitochondria)
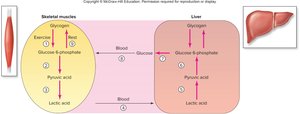
Interconversion of Glucose, Lactic Acid, and Glycogen
Glycogenesis and Glycogenolysis
Cells cannot store free glucose due to osmotic effects; instead, glucose is stored as glycogen. Glycogenesis is the synthesis of glycogen from glucose, primarily in the liver, skeletal muscle, and cardiac muscle. Glycogenolysis is the breakdown of glycogen to glucose-1-phosphate, which is converted to glucose-6-phosphate. Only the liver can release free glucose into the bloodstream due to the presence of glucose-6-phosphatase.
Glycogenesis: Formation of glycogen from glucose
Glycogenolysis: Breakdown of glycogen to glucose-1-phosphate
Locations: Liver, skeletal muscle, cardiac muscle
Why only liver releases glucose: Only liver has glucose-6-phosphatase
Cori Cycle and Gluconeogenesis
The Cori cycle describes the metabolic pathway in which lactic acid produced by muscles is transported to the liver, converted back to pyruvic acid, and then to glucose via gluconeogenesis. This glucose can be returned to the muscles for energy.
Gluconeogenesis: Synthesis of glucose from non-carbohydrate sources (e.g., pyruvic acid, amino acids)
Cori Cycle: Occurs between skeletal muscle and liver; recycles lactic acid to glucose
Metabolism of Lipids and Proteins
Lipid Metabolism: Lipogenesis and Lipolysis
When energy intake exceeds demand, glucose is converted to fatty acids and stored as triglycerides (lipogenesis). When energy is needed, triglycerides are broken down into fatty acids and glycerol (lipolysis). Fatty acids undergo β-oxidation to form acetyl CoA, which enters the citric acid cycle.
Lipogenesis: Formation of triglycerides from glucose and fatty acids
Lipolysis: Breakdown of triglycerides to fatty acids and glycerol
β-oxidation: Fatty acids are converted to acetyl CoA (e.g., 16-carbon fatty acid yields 8 acetyl CoA)
ATP Yield: High; a 16-carbon fatty acid can yield up to 108 ATP
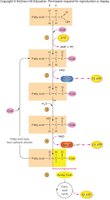
Ketogenesis
When fatty acid breakdown exceeds energy needs (e.g., fasting, diabetes), the liver converts excess acetyl CoA into ketone bodies (ketogenesis). These water-soluble molecules can be used as alternative energy sources but may accumulate and cause ketosis.
Amino Acid Metabolism and Gluconeogenesis
Amino acids are primarily used for protein synthesis, but excess can be converted to glucose (gluconeogenesis) or used for energy. Some citric acid cycle intermediates can be converted to amino acids by transamination (addition of an amine group).
Gluconeogenesis: Formation of glucose from amino acids
Transamination: Conversion of keto acids to amino acids
Integration of Metabolic Pathways
Carbohydrates, fats, and proteins are interconnected through shared metabolic intermediates, allowing the body to adapt to varying energy demands and dietary intake.
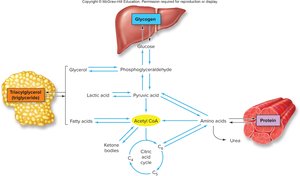
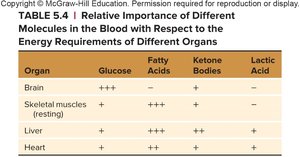
Relative Importance of Energy Sources in Different Organs
The preference for energy substrates varies by organ. For example, the brain relies heavily on glucose, while resting skeletal muscle prefers fatty acids. The liver and heart can utilize a variety of substrates, including ketone bodies and lactic acid.
Organ | Glucose | Fatty Acids | Ketone Bodies | Lactic Acid |
|---|---|---|---|---|
Brain | +++ | – | + | – |
Skeletal muscles (resting) | + | +++ | + | – |
Liver | ++ | ++ | + | + |
Heart | + | ++ | ++ | + |