 Back
BackThe Chemical Foundation of Life: Weak Interactions in an Aqueous Environment
Study Guide - Smart Notes

The Chemical Foundation of Life: Weak Interactions in an Aqueous Environment
Introduction
Biomolecules are stabilized and function through a complex interplay of covalent and noncovalent interactions. While covalent bonds provide the backbone for molecular structure, noncovalent interactions are essential for the dynamic processes and specific architectures observed in biological systems. This chapter explores the types, properties, and biological significance of weak interactions, especially in aqueous environments.
Noncovalent Interactions in Biochemistry
Importance and Examples
Noncovalent interactions are weak, reversible forces that play a critical role in the structure and function of biomolecules.
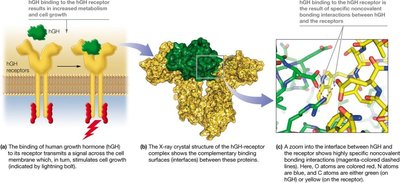
Examples include the binding of human growth hormone (hGH) to its receptor, the stabilization of DNA double helix, and the folding of proteins into their functional conformations.
These interactions allow for the dynamic assembly and disassembly of molecular complexes, essential for cellular processes.
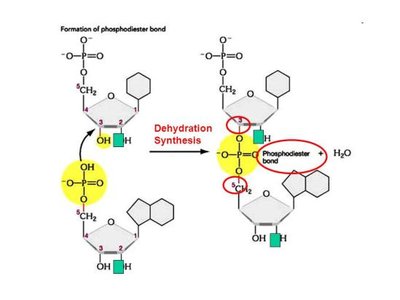
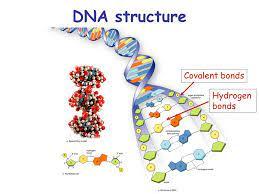
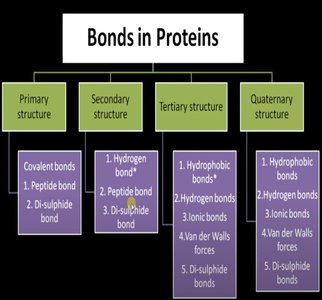
In DNA, the phosphodiester bond (covalent) maintains the linear sequence, while hydrogen bonds (noncovalent) stabilize the double helix.
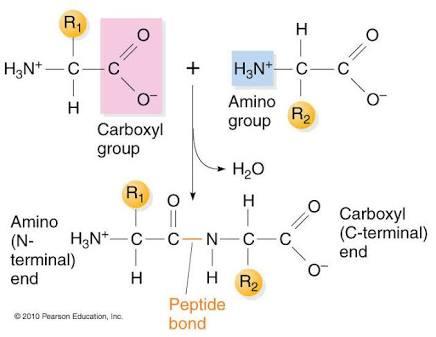
Proteins are composed of amino acids linked by peptide bonds (covalent), but their higher-order structures are stabilized by noncovalent interactions.
Covalent vs. Noncovalent Bond Energies
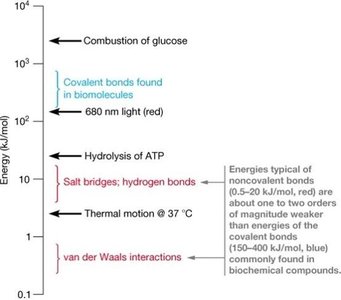
Covalent bonds have energies between 150–400 kJ/mol, providing stability to biomolecular backbones.
Noncovalent bonds are 10–100 times weaker (0.4–21 kJ/mol), allowing for reversible interactions necessary for biological function.
Their weakness is crucial for the dynamic nature of life, enabling continuous breakage and reformation.
The Nature and Types of Noncovalent Interactions
Overview of Noncovalent Interactions
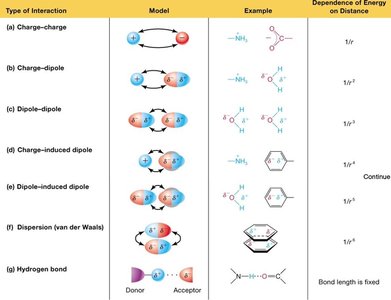
All noncovalent interactions are fundamentally electrostatic in nature.
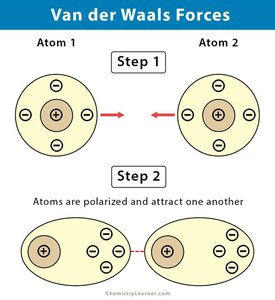
Types include charge–charge, charge–dipole, dipole–dipole, charge-induced dipole, dipole-induced dipole, van der Waals (dispersion), and hydrogen bonds.
Individually weak, but collectively can sum to significant energies in macromolecular assemblies.
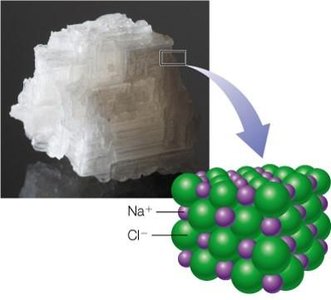
Charge–Charge Interactions (Salt Bridges)
Electrostatic interactions between two charged particles, described by Coulomb’s Law:
In biological systems, the force is reduced by the dielectric constant (ε) of the medium (water has ε ≈ 80):
Energy required to separate two charges:
Charge–charge interactions are important in protein–protein and protein–ligand binding, as well as in ionic crystals like NaCl.
Intermolecular Forces
Determine physical properties such as state of matter, melting/boiling points, and solubility.
Main types: hydrogen bonding, dipole–dipole interactions, and van der Waals forces.
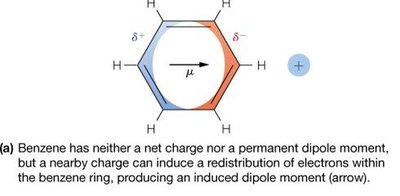
Dipole and Induced Dipole Interactions
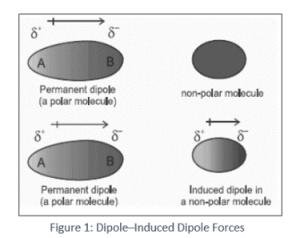
Polar molecules have a permanent dipole moment (μ), leading to dipole–dipole and charge–dipole interactions.
Nonpolar molecules can acquire an induced dipole in the presence of an electric field or nearby polar molecule.
Energy of interaction depends on orientation and distance (e.g., charge–dipole ∝ 1/r², dipole–dipole ∝ 1/r³).
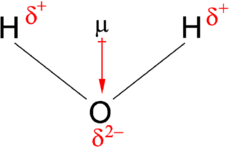
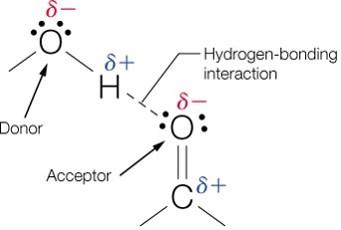
Hydrogen Bonding
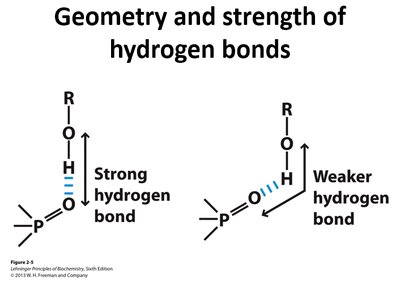
A hydrogen bond forms between a hydrogen atom covalently bonded to an electronegative atom (donor) and a lone pair on another electronegative atom (acceptor).
Hydrogen bonds are highly directional and strongest when the donor, hydrogen, and acceptor are colinear (180°).
They are crucial for the structure of DNA, proteins, and water.
The Role of Water in Biological Processes
Structure and Properties of Water
Water has two hydrogen bond donor sites, two acceptor sites, a permanent dipole, high heat capacity, high dielectric constant, and high surface tension.
These properties make water an ideal medium for life, supporting solubility, temperature regulation, and molecular interactions.
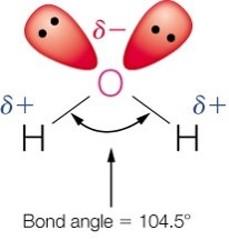
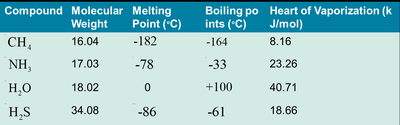
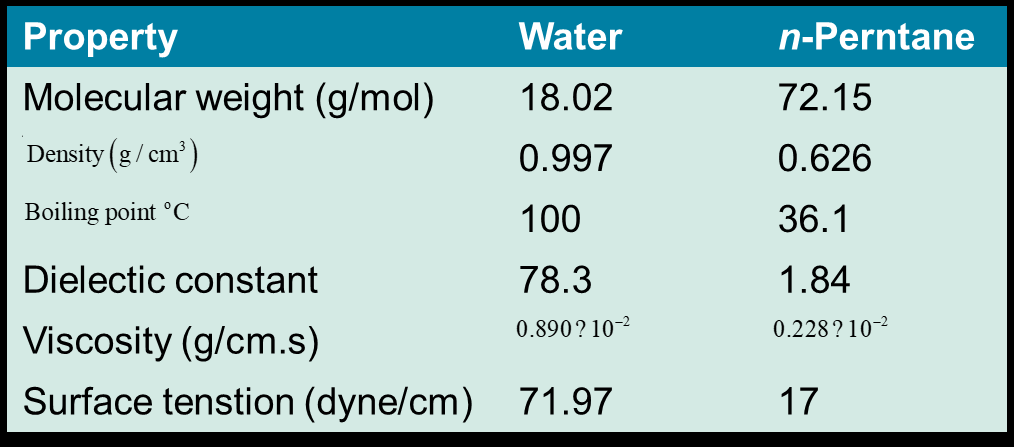
Physiological Relevance of Water
Water’s high heat of vaporization allows for effective cooling via perspiration.
Its high boiling point ensures it remains liquid at Earth’s surface temperatures.

Water as a Molecular Lattice
In ice, water forms a rigid tetrahedral lattice, making solid water less dense than liquid water (ice floats).
Liquid water retains a dynamic hydrogen-bonded network.
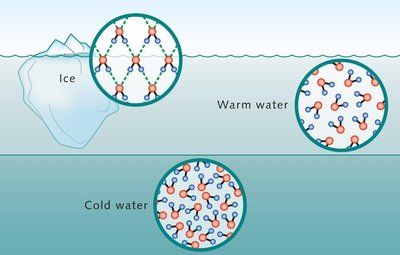
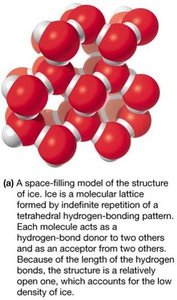
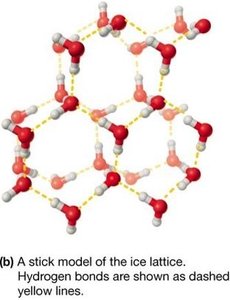
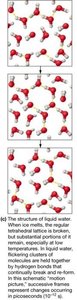
Solubility in Aqueous Solution
Hydrophilic Molecules
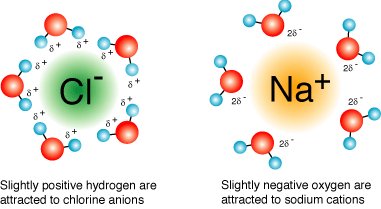
Hydrophilic substances dissolve readily in water due to their ability to form hydrogen bonds or interact with water’s dipole.
Ionic compounds are solvated by hydration shells, while polar molecules (e.g., proteins, nucleic acids) dissolve via hydrogen bonding.
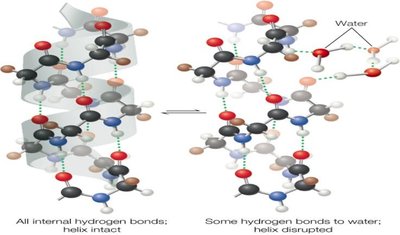
Hydrophobic Molecules and the Hydrophobic Effect
Hydrophobic molecules cannot form hydrogen bonds with water, leading to the formation of ordered water cages (clathrates) around them, which decreases entropy.
To minimize this unfavorable entropy loss, hydrophobic molecules aggregate, a phenomenon known as the hydrophobic effect.
This effect is critical for protein folding, DNA double helix formation, and lipid bilayer assembly.
Additional info: The hydrophobic effect is a major driving force in the self-assembly of biological membranes and macromolecular complexes.
Amphipathic Molecules
Amphipathic molecules contain both hydrophilic and hydrophobic regions, such as phospholipids in biological membranes.
They organize into structures like micelles and bilayers in water, essential for membrane formation.
Acid–Base Equilibria in Biochemistry
Acids, Bases, and Ionization
According to the Brønsted-Lowry definition, acids are proton donors and bases are proton acceptors.
Strong acids/bases dissociate completely; weak acids/bases dissociate partially.
Water can act as both an acid and a base, leading to the ion product of water ():
at 25°C
The pH Scale and Physiological pH
pH is defined as .
Most biological fluids have pH values between 6.5 and 8.0, the physiological pH range.
Buffer systems maintain pH stability, crucial for enzyme activity and metabolic processes.
Weak Acid and Base Equilibria
The strength of an acid is given by its dissociation constant ():
pKa is the negative logarithm of : .
The Henderson–Hasselbalch equation relates pH, pKa, and the ratio of conjugate base to acid:
Buffer Solutions and Clinical Relevance
Buffers resist changes in pH upon addition of acid or base, especially near their pKa values.
Major biological buffers include the dihydrogen phosphate system (intracellular) and the carbonic acid–bicarbonate system (blood).
Disruption of pH homeostasis can lead to acidemia or alkalemia, with severe physiological consequences.
Molecules with Multiple Ionizing Groups
Ampholytes and Titration Curves
Ampholytes are molecules with both acidic and basic groups (e.g., amino acids like glycine).
They exhibit complex titration behavior, with multiple pKa values and a characteristic isoelectric point (pI), where the net charge is zero.
For glycine:
Macroions and Electrostatic Interactions
Macroions in Solution
Polyelectrolytes (e.g., nucleic acids) and polyampholytes (e.g., proteins) are macroions whose solubility and interactions depend on pH and net charge.
Like-charged macroions repel, increasing solubility; oppositely charged macroions attract, leading to complex formation (e.g., DNA-histone complexes in chromatin).
