 Back
BackThe Role of Water in Living Systems
Study Guide - Smart Notes

The Role of Water in Living Systems
Introduction
Water is fundamental to all known forms of life, serving as the medium in which biochemical reactions occur and as a regulator of physiological processes. Its unique chemical and physical properties make it indispensable for maintaining the conditions necessary for life.
Water’s Unique Properties as the Medium of Life
Hydrogen Bonding and Molecular Structure
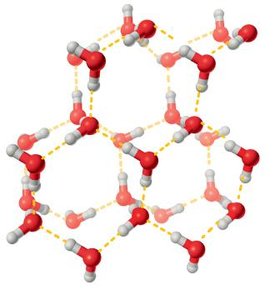
Water molecules are capable of forming up to four hydrogen bonds simultaneously, acting as both hydrogen bond donors and acceptors. This extensive hydrogen-bonding network is responsible for many of water’s remarkable properties.
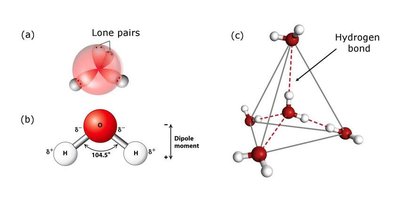
Tetrahedral Structure: Each water molecule has two hydrogen atoms and two lone pairs on the oxygen atom, resulting in a tetrahedral geometry.
Hydrogen Bonding: In ice, water molecules form a regular tetrahedral lattice, while in liquid water, this structure is transient but still significant.
Bond Energies: Hydrogen bonds in water have energies of 8.4–20.9 kJ/mol, much weaker than covalent bonds (209–418 kJ/mol), but their cumulative effect is substantial for molecular stability.
Physical Properties: Density, Melting and Boiling Points
Water exhibits unusual physical properties compared to other small molecules due to hydrogen bonding:
Density: Ice is less dense than liquid water, allowing it to float. This is due to the open, tetrahedral lattice structure in ice, which collapses partially in the liquid state, increasing density.
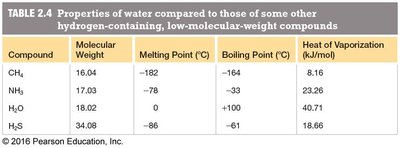
Melting and Boiling Points: Water has higher melting and boiling points than other hydrogen-rich compounds of similar molecular weight, due to strong intermolecular hydrogen bonds.
Polarity and Dielectric Constant
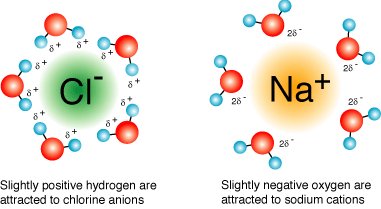
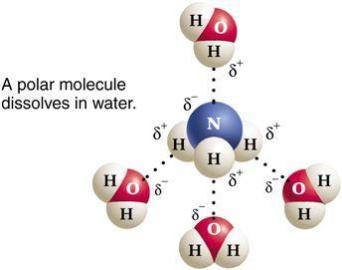
Water is a highly polar molecule, with a bond angle of 104.5° and a significant dipole moment. Its high dielectric constant (~80) makes it an excellent solvent for ionic and polar substances, as it can effectively weaken electrostatic interactions between ions.
Permanent Dipole: The oxygen atom is more electronegative than hydrogen, creating partial negative (δ−) and positive (δ+) charges.
Solvent Power: The high dielectric constant allows water to dissolve salts and polar molecules efficiently.
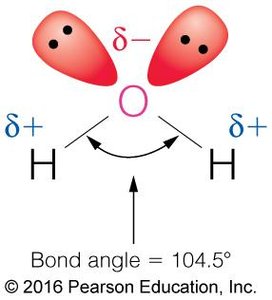
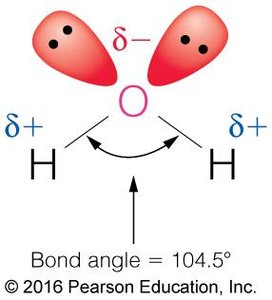
Thermal Properties: Heat Capacity and Temperature Regulation
Water has a high specific heat capacity (4187 J/kg°C), meaning it can absorb or release large amounts of heat with minimal temperature change. This property buffers organisms and environments against sudden temperature fluctuations, playing a major role in thermal regulation.
Heat of Vaporization: Water requires significant energy to change from liquid to gas, contributing to cooling mechanisms such as sweating.
Environmental Stability: Large bodies of water maintain nearly constant temperatures, supporting stable ecosystems.

Viscosity and Surface Tension
Water exhibits relatively high viscosity and surface tension compared to other liquids of similar molecular weight. Surface tension is the resistance of a liquid surface to distortion, which is crucial for processes such as capillary action in plants and the formation of droplets.
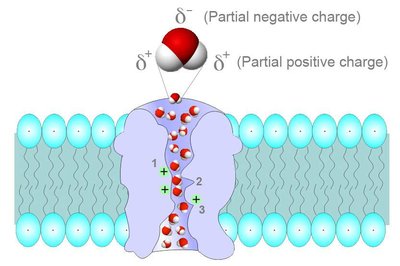
Water Transport in Biological Systems
Aquaporins and Membrane Permeability
Water crosses cell membranes primarily through specialized channel proteins called aquaporins. These channels selectively conduct water molecules while preventing the passage of ions and other solutes, maintaining osmotic balance and enabling rapid water movement.
Thermodynamic Equilibrium: Water concentration is balanced across membranes via aquaporins.
Biological Importance: Aquaporins are essential for processes such as kidney function and cellular hydration.
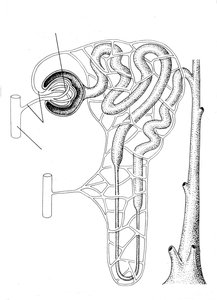
Water Reabsorption in the Kidneys
In the kidneys, water is reabsorbed from the nephron back into the bloodstream through aquaporin channels, particularly aquaporin 2. This process is regulated by the hormone vasopressin (antidiuretic hormone, ADH), which controls fluid balance by reducing urination.
Filtration: Blood is filtered in the glomerulus, and water is reabsorbed as needed.
Hormonal Regulation: Vasopressin increases water reabsorption, concentrating urine and conserving body water.
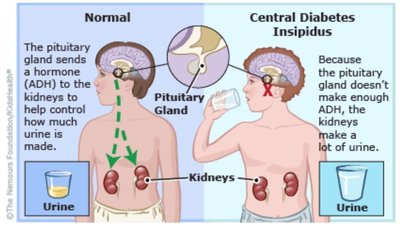
Disorders: Diabetes Insipidus
Diabetes insipidus is a disorder characterized by excessive loss of water in urine due to defects in aquaporin channels, vasopressin, or its receptor. This leads to an imbalance of body fluids and can cause dehydration if not managed properly.
Water as an Excellent Solvent
Dissolution of Ionic Compounds
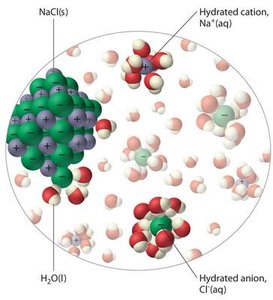
Water dissolves ionic compounds by hydrating ions, forming energetically favorable hydration shells. The high dielectric constant of water reduces the electrostatic attraction between oppositely charged ions, allowing them to disperse in solution.
Hydration Shells: Water molecules surround ions, stabilizing them in solution.
Biological Relevance: This property is essential for the function of electrolytes in cells and tissues.
Dissolution of Nonionic Polar Compounds
Nonionic polar compounds dissolve in water through hydrogen bonding or dipole-dipole interactions. Water’s polarity allows it to interact with and stabilize a wide range of biological molecules, including sugars and amino acids.
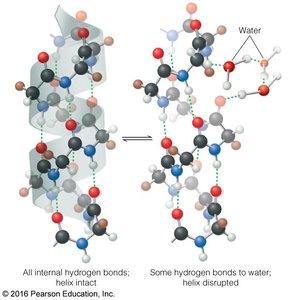
Solvation of Macromolecules
Water forms hydrogen bonds with solvent-accessible groups on proteins and nucleic acids, competing with intramolecular hydrogen bonds. This dynamic exchange is crucial for the structure and function of macromolecules in biological systems.
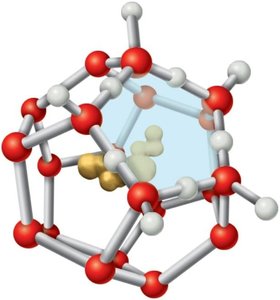
Hydrophobic Effect and Clathrate Structures
Water forms ordered "clathrate" cages around nonpolar (hydrophobic) molecules, decreasing the entropy of the solvent. When hydrophobic molecules aggregate, some water molecules are released from these cages, increasing the entropy of the system—a driving force for the hydrophobic effect, which is critical for membrane formation and protein folding.
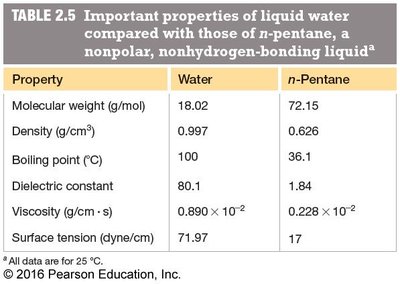
Summary Table: Key Properties of Water
Property | Water | n-Pentane |
|---|---|---|
Molecular weight (g/mol) | 18.02 | 72.15 |
Density (g/cm³) | 0.997 | 0.626 |
Boiling point (°C) | 100 | 36.1 |
Dielectric constant | 80.1 | 1.84 |
Viscosity (g/cm·s) | 0.890 × 10−2 | 0.228 × 10−2 |
Surface tension (dyne/cm) | 71.97 | 17 |

Key Equations
Dipole Moment:
Coulomb’s Law (force between ions):
Dielectric Constant Effect:
Additional info: The hydrophobic effect is a major driving force in the folding of proteins and the formation of biological membranes, as it minimizes the ordering of water molecules around nonpolar groups.
