 Back
BackThermodynamics and High-Energy Molecules in Biochemical Reactions
Study Guide - Smart Notes

Biochemical Reactions and Thermodynamics
Introduction to Biochemical Reactions
Biochemical reactions are chemical processes that occur within living cells, forming the basis of metabolism. These reactions are tightly regulated to ensure proper cellular function and energy flow.
Enzymes act as biological catalysts, increasing the rate of biochemical reactions by lowering activation energy.
Reactions are controlled both kinetically (by enzymes) and thermodynamically (by energy changes).
Metabolic pathways consist of sequences of biochemical reactions, each catalyzed by a specific enzyme.
Key Questions in Biochemical Reactions
Can a biological reaction occur? (Spontaneity, direction, thermodynamics)
How fast can it occur? (Reaction rate, kinetics)
Which enzymes are optimal targets for regulation or drug action?
Thermodynamics in Biological Systems
Basic Concepts and Laws
Thermodynamics is the study of energy transformations, particularly heat and work, and their relation to matter. It provides the framework for understanding the direction and feasibility of biochemical reactions.
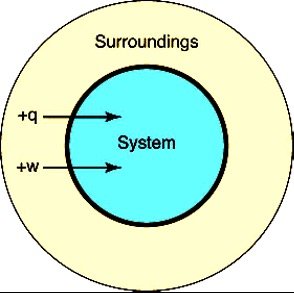
System: The part of the universe under study (e.g., a cell or reaction vessel).
Surroundings: Everything outside the system.

0th Law: If two systems are each in thermal equilibrium with a third, they are in equilibrium with each other.
1st Law: Energy is conserved. (change in internal energy equals heat added minus work done).
2nd Law: Entropy of the universe increases in any spontaneous process ().
3rd Law: As temperature approaches absolute zero, entropy approaches a minimum.
Key Thermodynamic Quantities
Internal Energy (U): Total energy contained within a system.
Enthalpy (H): ; reflects heat content at constant pressure.
Entropy (S): Measure of disorder or randomness.
Gibbs Free Energy (G): ; determines spontaneity of reactions.
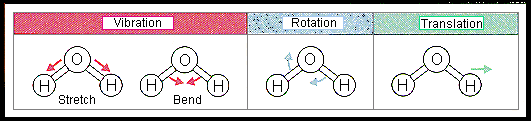
Translation, Vibration, and Rotation
Molecules possess different types of motion, each requiring energy:
Translation: Movement through space.
Vibration: Stretching and bending of bonds.
Rotation: Spinning around an axis.
Heat, Temperature, and Calorimetry
Heat (Q): Transfer of thermal energy due to temperature difference.
Temperature (T): Measure of average kinetic energy.
Calorimetry: Measurement of heat changes in chemical reactions.
Enthalpy Changes in Reactions
Exothermic: ; heat released to surroundings (e.g., catabolism).
Endothermic: ; heat absorbed from surroundings (e.g., anabolism).

Entropy and the Second Law
Entropy quantifies the degree of disorder in a system. The second law states that the total entropy of the universe increases in any spontaneous process.
Classic view:
Statistical view: , where is the number of possible microstates.
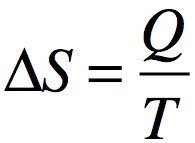
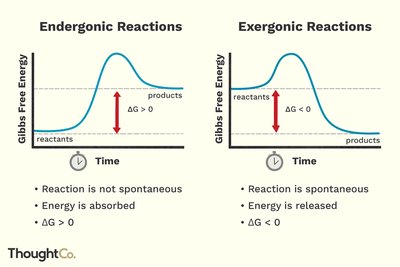
Gibbs Free Energy and Reaction Spontaneity
Gibbs free energy change () determines whether a reaction is spontaneous:
: Exergonic, spontaneous
: Endergonic, non-spontaneous
: Equilibrium
Equation:
Standard vs. Nonstandard Gibbs Free Energy
Standard Gibbs Free Energy (): Measured under standard conditions (25°C, 1 atm, 1 M concentrations, pH 7).
Nonstandard Gibbs Free Energy (): Actual cellular conditions; depends on concentrations of reactants and products.
Relationship:
= reaction quotient = [products]/[reactants]
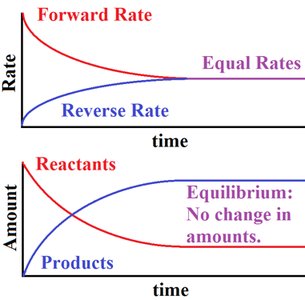
Equilibrium and Steady State
Equilibrium: Forward and reverse reaction rates are equal; .
Steady State: Concentrations of intermediates remain constant, but the system is not at equilibrium (typical in living cells).
Classification of Biological Reactions
Anabolism vs. Catabolism
Anabolism: Biosynthetic, endothermic reactions; require energy input (e.g., protein synthesis).
Catabolism: Degradative, exothermic reactions; release energy (e.g., glycolysis).
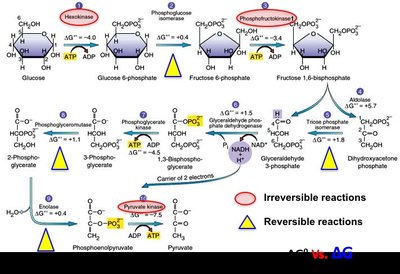
Metabolically Reversible vs. Irreversible Reactions
Metabolically reversible: near zero; direction depends on substrate/product concentrations; common in central metabolism.
Metabolically irreversible: ; cannot reverse under cellular conditions; often regulatory points and drug targets.
High-Energy Molecules in Metabolism
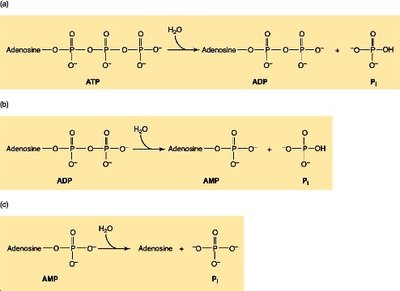
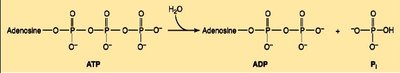
ATP: The Universal Energy Currency
Adenosine triphosphate (ATP) stores and transfers energy via high-energy phosphate bonds.
Hydrolysis of ATP to ADP or AMP releases significant free energy ( ≈ –30 to –32 kJ/mol).
ATP hydrolysis is coupled to endergonic reactions to drive them forward.
Mechanisms of ATP Hydrolysis
Increase in entropy (more product molecules, more disorder).
Relief of electrostatic repulsion between phosphate groups.
Greater resonance stabilization in inorganic phosphate (Pi) than in ATP.
Energy Coupling in Metabolism
Endergonic reactions are coupled to ATP hydrolysis to make the overall process exergonic.
Example: Asparagine synthetase couples asparagine formation (endergonic) with ATP hydrolysis (exergonic).
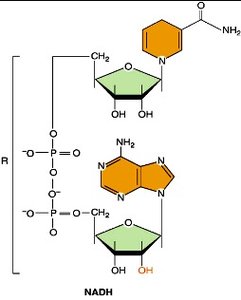
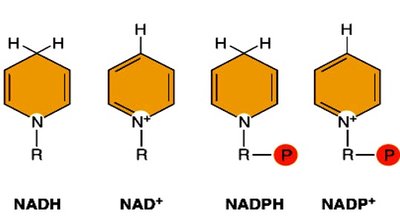
NADH and NADPH: Electron Carriers
NADH: Produced in catabolic pathways (glycolysis, Krebs cycle); donates electrons to the electron transport chain for ATP synthesis.
NADPH: Produced in the pentose phosphate pathway and photosynthesis; used in biosynthetic (anabolic) reactions and antioxidant defense.
Both are derived from niacin (vitamin B3).
Major Pathways for High-Energy Molecule Generation
ATP: Generated mainly via cellular respiration (glycolysis, Krebs cycle, oxidative phosphorylation).
NADH: Produced during glycolysis and the Krebs cycle; used to generate ATP in the electron transport chain.
NADPH: Generated mainly via the pentose phosphate pathway; used in lipid and nucleotide biosynthesis.
Metabolic Pathways and Cofactors
Metabolic Pathway Structure
Metabolic pathways are sequences of enzyme-catalyzed reactions where the product of one reaction becomes the substrate for the next.
Pathways can be anabolic (building up) or catabolic (breaking down).
Mobile vs. Bound Cofactors
Mobile cofactors (co-substrates): Transiently bind to enzymes and transfer functional groups or electrons (e.g., ATP, NADH, NADPH).
Prosthetic groups (bound cofactors): Permanently attached to enzymes (e.g., heme in hemoglobin).
Pathway Flux
Pathway flux is the overall rate of a metabolic pathway, determined by the concentrations of substrates and end-products.
Regulation often occurs at key irreversible steps (rate-limiting enzymes).
Summary Table: Classification of Biological Reactions
Classification | Energy Change | Thermodynamic Feature | Example |
|---|---|---|---|
Anabolic | Endothermic () | Requires energy input | Protein synthesis |
Catabolic | Exothermic () | Releases energy | Glycolysis |
Metabolically reversible | Direction depends on [substrate]/[product] | Most central metabolic steps | |
Metabolically irreversible | Unidirectional, regulatory | Hexokinase in glycolysis |
