 Back
BackWater: Physical and Chemical Properties Essential for Life
Study Guide - Smart Notes

Water as the Medium for Life
Importance of Water in Biological Systems
Water is the fundamental medium in which life originated and continues to thrive. Early cells formed in water, enclosing a small volume within a membrane, allowing solutes to dissolve and chemical reactions to occur efficiently.
Cellular Environment: Most of a cell and its organelles are composed of water, forming the cytoplasm where metabolic reactions take place.
Transport Medium: Water facilitates the transport of substances into and out of cells, and in multicellular organisms, it is found as interstitial fluid between cells.
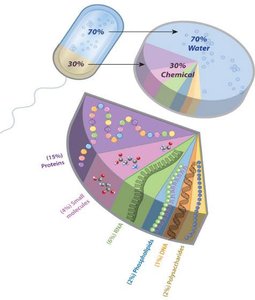
Proportion in Organisms: Over 70% of Earth's surface is water, and living organisms are largely composed of water.

Water’s Polarity and Hydrogen Bonding
Structure and Properties of Water Molecules
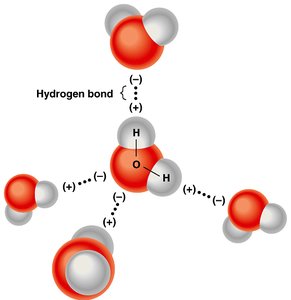
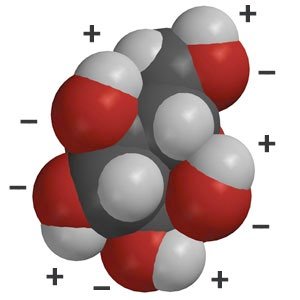
Water molecules possess polar covalent bonds due to unequal electron sharing between oxygen and hydrogen atoms. The oxygen atom, with a larger nucleus, attracts shared electrons more strongly, resulting in partial charges: δ+ on hydrogen and δ- on oxygen.
Polarity: The unequal sharing of electrons creates a dipole moment, making water a polar molecule.
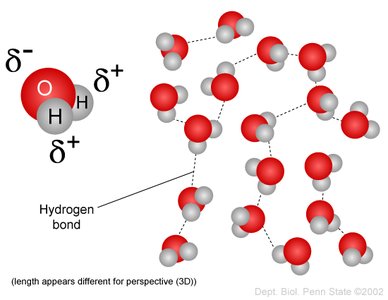
Hydrogen Bonding: The partial charges enable weak hydrogen bonds between adjacent water molecules, which collectively provide significant strength.
Cohesion and Consequences for Organisms
Surface Tension and Biological Implications
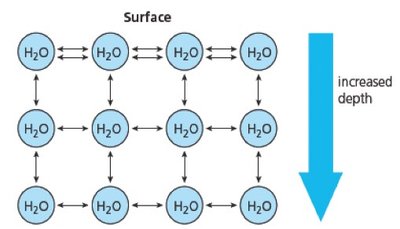
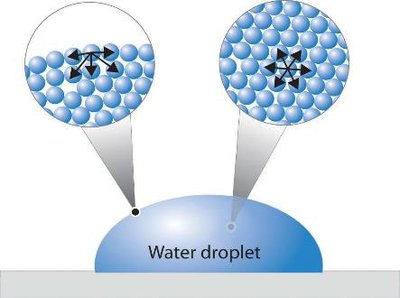
Cohesion refers to the attraction between water molecules due to hydrogen bonding. This property leads to surface tension, allowing water to form droplets and providing habitats for certain organisms.
Surface Tension: Water molecules at the surface form stronger bonds with their neighbors, resulting in inward contraction and droplet formation.
Biological Habitats: Surface tension supports insects like water striders and mosquito larvae, enabling them to live on or under the water surface.
Adhesion and Its Impact on Living Systems
Capillary Action and Water Transport
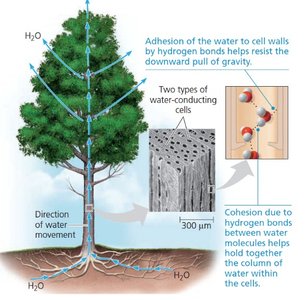
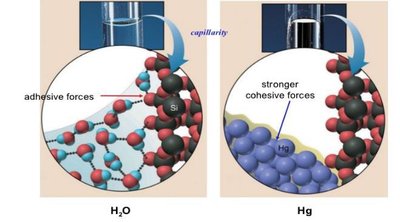
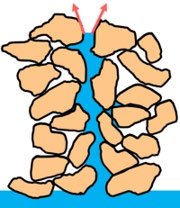
Adhesion is the attraction between water molecules and polar surfaces, such as cell walls or glass. This property enables capillary action, which is essential for water transport in plants and soil.
Capillary Action: Water is drawn through narrow tubes (e.g., xylem vessels) due to adhesion, allowing plants to absorb water from soil.
Meniscus Formation: Adhesive forces between water and glass create a meniscus in test tubes, unlike nonpolar liquids like mercury.
Water as a Solvent in Metabolism and Transport
Solubility and Biological Reactions
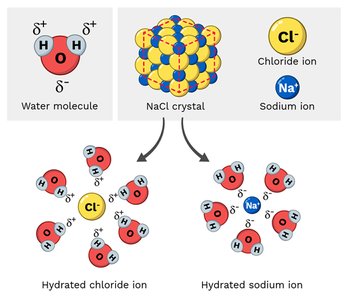
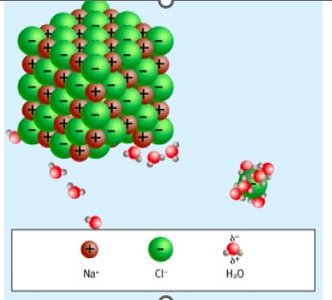
Water’s polarity makes it an excellent solvent for hydrophilic molecules, enabling metabolic reactions and transport of solutes. Hydrophobic molecules behave differently, often forming separate phases.
Hydrophilic Substances: Polar and charged molecules (e.g., salts, amino acids, glucose) dissolve well in water, facilitating metabolic and enzymatic reactions.
Hydrophobic Substances: Nonpolar molecules (e.g., lipids, fats, cholesterol) are insoluble in water and dissolve in nonpolar solvents.
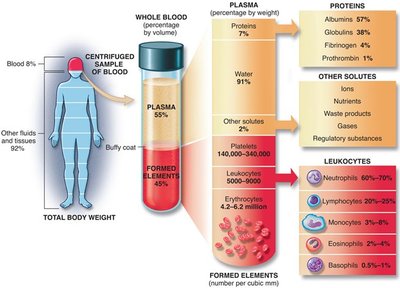
Blood Plasma: 91% of blood plasma is water, making solubility crucial for effective transport in the circulatory system.

Physical Properties of Water and Their Effects on Aquatic Animals
Buoyancy, Viscosity, Thermal Conductivity, and Specific Heat Capacity
Water’s unique physical properties influence the survival and adaptation of aquatic organisms.
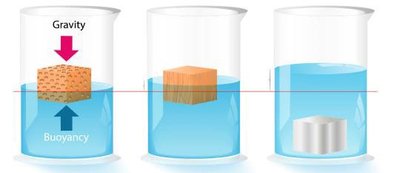
Buoyancy: Water exerts an upward force on objects, allowing organisms with densities close to water to float easily.
Viscosity: Water’s low viscosity compared to other liquids enables efficient movement for aquatic animals.
Thermal Conductivity: Water conducts heat efficiently, requiring aquatic animals to adapt to prevent heat loss.
Specific Heat Capacity: Water’s high specific heat capacity stabilizes temperature, providing a suitable habitat for organisms sensitive to temperature changes.
Comparison Table: Solubility of Substances in Water
Solubility and Chemical Properties
Molecule | Chemical Property | Soluble/insoluble in blood/water |
|---|---|---|
Glucose | Polar due to hydroxyl groups (OH-groups) which cause water molecules to form hydrogen bonds with it. | Soluble |
Amino Acids | Solubility depends on the R-group. Some are charged, some polar, some nonpolar. All are soluble to a certain degree due to zwitterion formation. | Mostly soluble |
Cholesterol | Mostly nonpolar, except for one OH group. Not sufficiently polar for solubility. | Insoluble |
Fats & lipids | Entirely non-polar | Insoluble |
Oxygen | Non-polar; solubility depends on temperature (better in cold water). | Very weakly soluble |
Sodium chloride | Polar molecule due to cations (Na+) & anion (Cl-) held together by ionic bond. Ions are surrounded by polar water molecules when dissolved. | Soluble |
Key Equations
Buoyancy
Buoyant Force:
Specific Heat Capacity:
Summary
Water’s physical and chemical properties—polarity, hydrogen bonding, cohesion, adhesion, solvent abilities, and unique physical characteristics—are essential for life. These properties enable metabolic reactions, transport, and adaptation in diverse biological systems, making water indispensable in biochemistry and cellular biology.
