 Back
BackOrigins of Atoms, Nuclear Chemistry, and Chemical Bonding: Foundations of General Chemistry
Study Guide - Smart Notes

Elements, Atoms, and the Law of Chemical Combinations
Origins of Atoms in the Universe
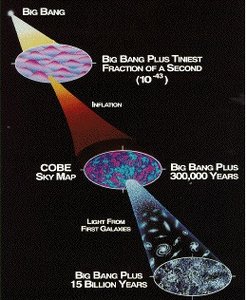
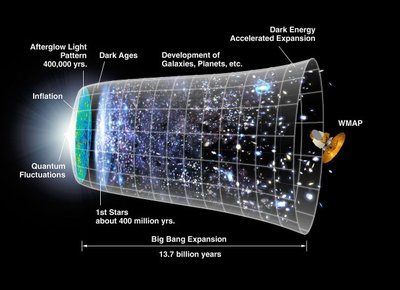
The atoms that make up all matter, including those in our bodies, originated from processes that began with the Big Bang. Understanding the formation and distribution of atoms is fundamental to chemistry and cosmology.
Big Bang Theory: The universe began approximately 13.8 billion years ago from a singularity, leading to the creation of fundamental particles and, eventually, atoms.
Formation of Elements: In the first few minutes after the Big Bang, the universe cooled enough for protons and neutrons to combine, forming hydrogen, helium, and trace amounts of lithium.
Stellar Nucleosynthesis: Heavier elements were formed later in stars through nuclear fusion processes.
Evidence: The expansion of the universe (red shift), cosmic microwave background radiation, and the observed abundance of light elements support these theories.
Law of Chemical Combinations: Atoms combine in fixed ratios to form compounds, reflecting their discrete and countable nature.
Nuclear Chemistry
Chemical vs. Nuclear Reactions
Chemical and nuclear reactions differ fundamentally in the particles involved and the changes that occur.
Chemical Reactions: Involve rearrangement of valence electrons; the identity of the element does not change.
Nuclear Reactions: Involve changes in the nucleus, often resulting in the transformation of one element into another.
Types of Nuclear Reactions
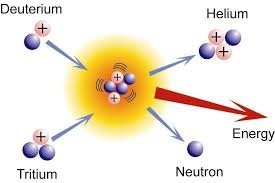
Fusion: Two light nuclei combine to form a heavier nucleus, releasing energy. Example: Fusion in stars forms helium from hydrogen.
Fission: A heavy nucleus splits into lighter nuclei, releasing energy and neutrons. Used in nuclear reactors and atomic bombs.
Radioactive Decay: Unstable nuclei emit particles (alpha, beta, gamma) to become more stable.
Forces in the Nucleus
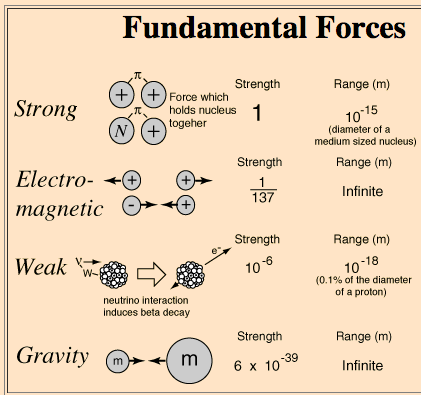
Four fundamental forces govern interactions in the universe, but only the strong nuclear force and electromagnetic force are significant in nuclear chemistry.
Strong Nuclear Force: Holds protons and neutrons together in the nucleus; strongest force but acts over very short distances (~10-15 m).
Electromagnetic Force: Causes repulsion between positively charged protons; acts over infinite range but is weaker than the strong force at nuclear distances.
Weak Nuclear Force: Responsible for certain types of radioactive decay (e.g., beta decay).
Gravity: Negligible at atomic scales.
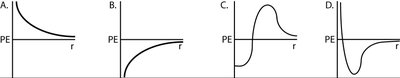
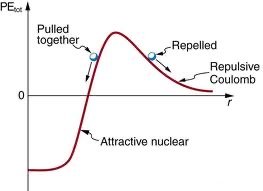
Potential Energy in Nuclear Interactions
As two nuclei approach, their potential energy changes due to the interplay of repulsive and attractive forces.
At large distances, electrostatic repulsion dominates, increasing potential energy as nuclei approach.
At very short distances, the strong nuclear force becomes attractive and can overcome repulsion, leading to fusion if enough energy is supplied.
Fusion in Stars and on Earth
Fusion powers stars, including our Sun, by converting hydrogen into helium and releasing vast amounts of energy.
Fusion Reaction in the Sun:
On Earth, achieving the necessary conditions for fusion (high temperature and pressure) is technologically challenging.

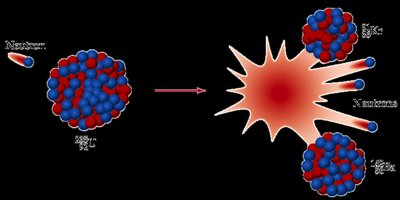
Nuclear Fission
Fission is the splitting of a heavy nucleus into lighter nuclei, releasing energy and neutrons, which can sustain a chain reaction.
Critical Mass: The minimum amount of fissile material needed to maintain a self-sustaining chain reaction.
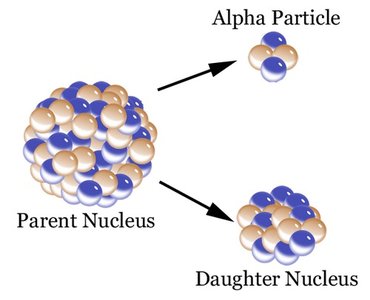
Radioactivity and Types of Decay
Radioactive decay is the process by which unstable nuclei emit particles or energy to become more stable.
Alpha Decay: Emission of an alpha particle ().
Beta Decay: Emission of a beta particle (electron or positron).
Gamma Decay: Emission of high-energy electromagnetic radiation.
Electron Capture: The nucleus captures an inner electron, converting a proton to a neutron.
Chemical Bonds, Molecular Structures, and Theories of Chemical Bonding
Why Do Atoms Form Bonds?
Atoms form bonds to achieve lower potential energy and greater stability. The nature of these bonds determines the properties of substances.
Covalent Bonds: Atoms share electrons, resulting in a stable arrangement and the release of energy.
Bond Length: The most stable distance between two bonded atoms, corresponding to the minimum potential energy.
Molecular Orbital (MO) Theory
MO theory describes how atomic orbitals combine to form molecular orbitals, which can be bonding or antibonding.
Bonding Orbitals: Lower energy, stabilize the molecule.
Antibonding Orbitals: Higher energy, destabilize the molecule.
Electrons fill the lowest energy orbitals first (Aufbau principle).
Bonding in Metals and Band Theory
In metals, atomic orbitals combine to form bands of molecular orbitals, allowing electrons to move freely and giving rise to metallic properties.
Conductivity: Free electrons allow metals to conduct electricity and heat.
Malleability: Atoms can move past each other without breaking bonds.
Shininess: Metals absorb and re-emit photons, giving them a shiny appearance.
Band Theory: In metals, the valence band overlaps with the conduction band; in semiconductors, a small gap exists; in insulators, the gap is large.
Valence Bond Theory and Hybridization
Valence Bond Theory explains bonding as the overlap of atomic orbitals, which may hybridize to form new orbitals for bonding.
Hybridization: Atomic orbitals mix to form hybrid orbitals (e.g., sp3 in diamond, sp2 in graphite).
Diamond: Each carbon forms four sp3 hybridized bonds in a tetrahedral geometry, resulting in a hard, non-conductive structure.
Graphite: Each carbon forms three sp2 hybridized bonds in a planar structure, with delocalized pi electrons allowing electrical conductivity.
Comparison of Covalent Bonds and London Dispersion Forces (LDF)
Covalent Bonds: Strong, directional, within molecules or networks, require significant energy to break.
LDF (van der Waals): Weak, due to temporary fluctuations in electron distribution, present between all molecules, increase with size of electron cloud.
Allotropes of Carbon: Diamond vs. Graphite
Property | Diamond | Graphite |
|---|---|---|
Structure | 3D network (tetrahedral) | 2D sheets (planar) |
Bonding | sp3 hybridized, sigma bonds | sp2 hybridized, sigma and delocalized pi bonds |
Hardness | Very hard | Soft, slippery |
Electrical Conductivity | Insulator | Conductor |
Melting Point | Very high (decomposes) | High |
Thermochemistry and Thermodynamics
Energy Changes in Chemical and Physical Processes
Energy is involved in breaking and forming bonds, as well as in changes of state (solid, liquid, gas).
Bond Formation: Releases energy to the surroundings.
Bond Breaking: Requires energy input.
Phase Changes: Melting and boiling require energy to overcome intermolecular forces; condensation and freezing release energy.
Summary
This guide covers the origins of atoms, the differences between chemical and nuclear reactions, the forces and energy changes involved in nuclear processes, and the fundamental theories of chemical bonding. Understanding these concepts is essential for further study in general chemistry.
