Explain why [CoCl4]2- (blue) and [Co(H2O)6]2+ (pink) have different colors. Which complex has its absorption bands at longer wavelengths?
Ch.21 - Transition Elements and Coordination Chemistry

Chapter 21, Problem 21.30
Classify the following ligands as monodentate, bidentate, tri-dentate, or tetradentate. Which can form chelate rings?
(a)
(b)
(c)
(d)
 Verified step by step guidance
Verified step by step guidance1
1. To classify ligands, we need to know their structures and how many sites they can use to bind to a central metal ion. Monodentate ligands have one site, bidentate ligands have two, tridentate ligands have three, and tetradentate ligands have four.
2. A ligand can form a chelate ring if it is at least bidentate, meaning it has two or more sites to bind to the central metal ion. This allows it to 'wrap around' the metal ion and form a ring structure.
3. Unfortunately, without the specific ligands provided in the problem, we cannot classify them or determine if they can form chelate rings. Please provide the specific ligands for further assistance.
4. Once the ligands are provided, you can identify the number of potential binding sites by looking for atoms with lone pairs of electrons. These are typically oxygen, nitrogen, or sulfur atoms in the ligand.
5. To determine if a ligand can form a chelate ring, look for multiple binding sites that are close enough together on the ligand to both bind to the same metal ion. If this is possible, the ligand can form a chelate ring.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Was this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Ligand Denticity
Denticity refers to the number of donor atoms in a ligand that can bind to a central metal atom in a coordination complex. Monodentate ligands have one donor atom, bidentate ligands have two, tridentate have three, and tetradentate have four. Understanding denticity is crucial for classifying ligands and predicting their behavior in coordination chemistry.
Recommended video:
Guided course

Ligands Example
Chelation
Chelation is the process by which a multidentate ligand binds to a metal ion, forming a stable ring structure known as a chelate. This occurs when the ligand wraps around the metal ion, creating a more stable complex than with monodentate ligands. Recognizing which ligands can form chelate rings is essential for understanding their stability and reactivity in various chemical environments.
Recommended video:
Guided course
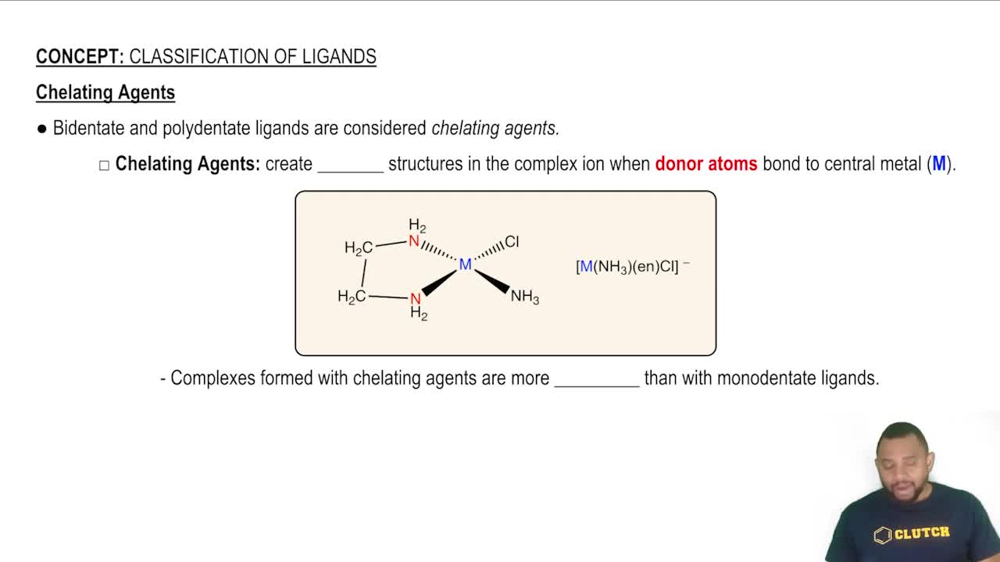
Chelating Agents
Coordination Complexes
Coordination complexes consist of a central metal atom bonded to surrounding ligands. The nature of these ligands, including their denticity, influences the geometry, stability, and reactivity of the complex. Familiarity with coordination chemistry is necessary to classify ligands and predict the formation of chelate rings in these complexes.
Recommended video:
Guided course
Coordination Complexes Example
Related Practice
Textbook Question
117
views
1
rank
Textbook Question
What is the crystal field energy level diagram for the complex [Fe(NH3)6]3+?
(a)
(b)
(c)
(d)
168
views
Textbook Question
Draw a crystal field energy-level diagram, assign the electrons to orbitals, and predict the number of unpaired electrons for each of the following.
(a) [Cu(en)3]2+
(b) [FeF6]2-
(c) [Co(en)3]3+ (low spin)
93
views
Textbook Question
For each of the following complexes, draw a crystal field energy-level diagram, assign the electrons to orbitals, and predict the number of unpaired electrons.
(d) [Cu(en)2]2+ (square planar)
118
views
Textbook Question
What is the highest oxidation state for each of the elements from Sc to Zn?
101
views
Textbook Question
What is the general trend in standard potentials for the oxidation of first-series transition metals from Sc to Zn? What is the reason for the trend?
95
views
