Classify the following ligands as monodentate, bidentate, tri-dentate, or tetradentate. Which can form chelate rings?
(a)
(b)
(c)
(d)

 Verified step by step guidance
Verified step by step guidance


Classify the following ligands as monodentate, bidentate, tri-dentate, or tetradentate. Which can form chelate rings?
(a)
(b)
(c)
(d)
For each of the following complexes, draw a crystal field energy-level diagram, assign the electrons to orbitals, and predict the number of unpaired electrons.
(d) [Cu(en)2]2+ (square planar)
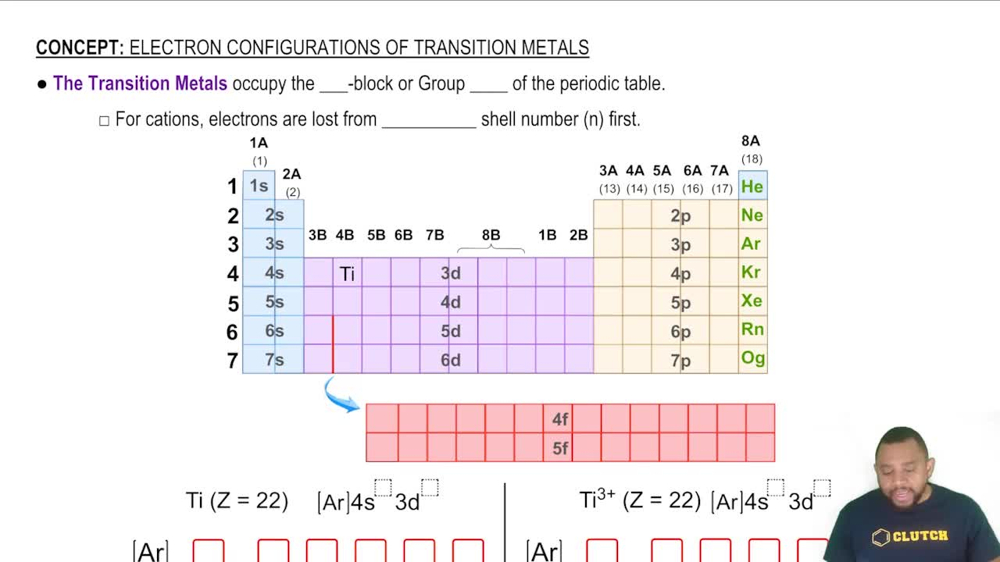
What is the highest oxidation state for each of the elements from Sc to Zn?
Draw a crystal field energy-level diagram for a square planar complex, and explain why square planar geometry is especially common for d8 complexes.
Predict the crystal field energy-level diagram for a square pyramidal ML5 complex that has two ligands along the axes but only one ligand along the z axis. Your diagram should be intermediate between those for an octahedral ML6 complex and a square planar ML4 complex.
What is a racemic mixture? Does it affect plane-polarized light? Explain.