What is a racemic mixture? Does it affect plane-polarized light? Explain.

 McMurry 8th Edition
McMurry 8th Edition Ch.21 - Transition Elements and Coordination Chemistry
Ch.21 - Transition Elements and Coordination Chemistry Problem 21.120
Problem 21.120Look at the colors of the isomeric complexes in Figure 21.12, and predict which is the stronger field ligand, nitro (-NO2) of nitrito (-ONO). Explain.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Ligand Field Theory

Crystal Field Splitting
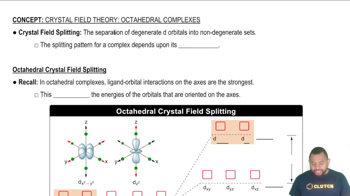
Field Strength of Ligands

The amount of paramagnetism for a first-series transition metal complex is related approximately to its spin-only magnetic moment. The spin-only value of the magnetic moment in units of Bohr magnetons (BM) is given by sqrt(n(n + 2)), where n is the number of unpaired electrons. Calculate the spin-only value of the magnetic moment for the 2+ ions of the first-series transition metals (except Sc) in octahedral complexes with (a) weak-field ligands and (b) strong-field ligands. For which electron configurations can the magnetic moment distinguish between high-spin and low-spin electron configurations?
The oxalate ion is a bidentate ligand as indicated in Figure 21.8. Would you expect the carbonate ion to be a monodentate or bidentate ligand? Explain your reasoning.
Draw the structures of all possible diastereoisomers of an octahedral complex with the formula MA2B2C2. Which of the diastereoisomers, if any, can exist as enantiomers?
Which of the following complexes are paramagnetic?
(a) [Mn(CN)6]3-
(b) [Zn(NH3)4]2+ (tetrahedral)
(c) [Fe(CN)6]4-
(d) [FeF6]4-
Based on the wavelength of maximum absorption of the cobalt complexes, arrange the following ligands in a spectrochemical series from weakest-field to strongest-field ligand.
(a) Cl- < NCS- < H2O < NH3
(b) Cl- < NCS- < H2O < NH3
(c) H2O < Cl- < NH3 < NCS-
(d) Cl- < H2O < NCS- < NH3