Back
BackCarbohydrates: Structure, Reactions, and Biological Roles
Study Guide - Smart Notes

Carbohydrates: Structure, Reactions, and Biological Roles
Ring Formation in Carbohydrates
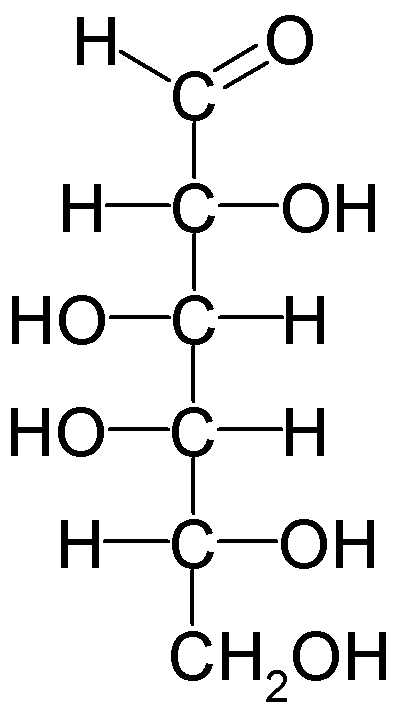
Carbohydrates can exist in both linear and cyclic (ring) forms. The ring structure is especially important for monosaccharides such as glucose and fructose. In ketose sugars, the anomeric carbon is typically C2, and the orientation of the hydroxyl group at this carbon determines the α (alpha) or β (beta) anomer.
Anomeric Carbon: The carbon derived from the carbonyl group (aldehyde or ketone) during ring closure; its configuration (α or β) is crucial for sugar properties.
α-Anomer: The OH group on the anomeric carbon is trans (opposite side) to the CH2OH group.
β-Anomer: The OH group on the anomeric carbon is cis (same side) to the CH2OH group.
Example: D-fructose forms a five-membered ring (furanose) with the anomeric carbon at C2.
Oxidation–Reduction and Reducing Sugars
Carbohydrates can undergo oxidation and reduction reactions, which are important in metabolism and analytical chemistry. A reducing sugar is one that can donate electrons to another molecule, typically by having a free aldehyde or ketone group.
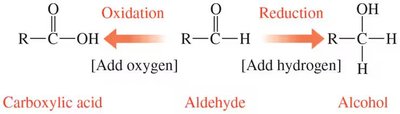
Oxidation: Involves the addition of oxygen or removal of hydrogen, converting an aldehyde to a carboxylic acid.
Reduction: Involves the addition of hydrogen, converting an aldehyde to an alcohol.
Reducing Sugars: Sugars with a free anomeric carbon that can reduce mild oxidizing agents (e.g., glucose).
Example: D-glucose is a reducing sugar because its anomeric carbon can be oxidized to a carboxylic acid.
Disaccharides: Formation and Glycosidic Bonds
Disaccharides are formed by the condensation (dehydration synthesis) of two monosaccharides, creating a glycosidic bond. Hydrolysis is the reverse reaction, breaking the bond with water.
Condensation Reaction: Two monosaccharides join, releasing water and forming a glycosidic bond.
Hydrolysis: Addition of water breaks a glycosidic bond, yielding two monosaccharides.
Glycosidic Bond: The covalent bond formed between the anomeric carbon of one sugar and a hydroxyl group of another.
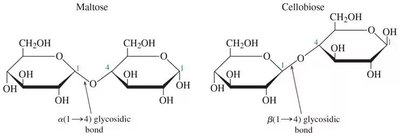
Naming Glycosidic Bonds: Indicate the carbon numbers and the α or β configuration (e.g., α(1→4) or β(1→4)).
Example: Maltose has an α(1→4) glycosidic bond; cellobiose has a β(1→4) glycosidic bond.
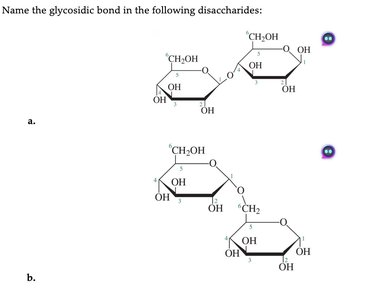
Common Disaccharides
Three important disaccharides are maltose, lactose, and sucrose, each formed from specific monosaccharide units and glycosidic linkages.
Maltose: Composed of two D-glucose units joined by an α(1→4) glycosidic bond.
Lactose: Composed of D-galactose and D-glucose joined by a β(1→4) glycosidic bond.
Sucrose: Composed of D-glucose and D-fructose joined by an α,β(1→2) glycosidic bond.
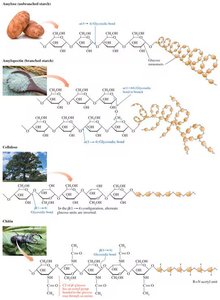
Polysaccharides: Structure and Function
Polysaccharides are long chains of monosaccharide units linked by glycosidic bonds. They serve as energy storage (starch, glycogen) or structural components (cellulose, chitin).
Starch: Storage polysaccharide in plants; consists of amylose (unbranched) and amylopectin (branched).
Cellulose: Structural polysaccharide in plants; composed of β(1→4) linked glucose units, forming rigid fibers.
Chitin: Structural polysaccharide in fungi and arthropods; composed of N-acetylglucosamine units.
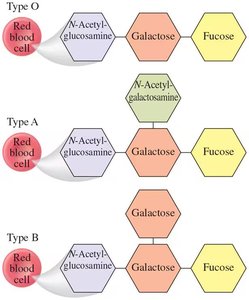
Blood and Carbohydrates
Carbohydrates play a crucial role in determining blood types through specific oligosaccharide structures on the surface of red blood cells. These structures are recognized by the immune system and are the basis for blood group compatibility.
Blood Group Antigens: Oligosaccharide chains attached to proteins and lipids on red blood cells determine blood type (A, B, AB, O).
Type O: Has the basic oligosaccharide structure.
Type A: Has an additional N-acetylgalactosamine.
Type B: Has an additional galactose.
Blood Transfusion Compatibility: Determined by the presence or absence of these carbohydrate antigens.
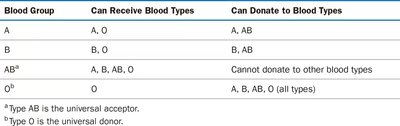
Blood Group Compatibility Table
Blood Group | Can Receive Blood Types | Can Donate to Blood Types |
|---|---|---|
A | A, O | A, AB |
B | B, O | B, AB |
ABa | A, B, AB, O | Cannot donate to other blood types |
Ob | O | A, B, AB, O (all types) |
Additional info: Type AB is the universal acceptor; type O is the universal donor.