 Back
BackCarboxylic Acids and Their Derivatives: Properties, Nomenclature, Reactions, and Biological Importance
Study Guide - Smart Notes

Carboxylic Acids and Their Derivatives
Overview
Carboxylic acids and their derivatives are fundamental organic compounds with significant roles in both chemistry and biochemistry. This chapter explores their structures, properties, nomenclature, reactions, and biological relevance.
Properties and Structures of Carboxylic Acids, Esters, and Amides
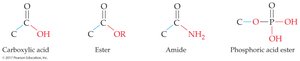
Functional Groups
Carboxylic acids, esters, and amides share a carbonyl group (C=O) but differ in the group attached to the carbonyl carbon:
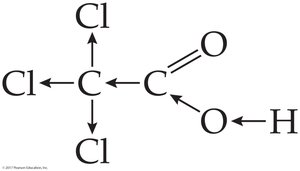
Carboxylic acid: RCOOH (carbonyl bonded to –OH)
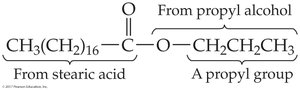
Ester: RCOOR' (carbonyl bonded to –OR')
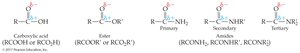
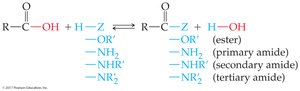
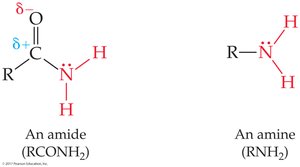
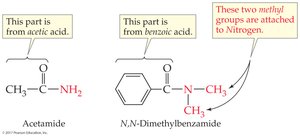
Amide: RCONH2, RCONHR', or RCONR'2 (carbonyl bonded to nitrogen)
Physical Properties
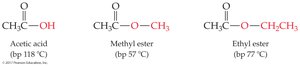
All boil at higher temperatures than comparable alkanes due to polarity.
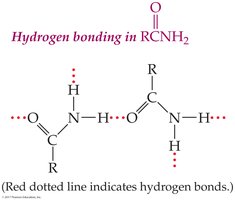
Carboxylic acids and amides (with H on N) can hydrogen bond, increasing boiling points and solubility.
Esters are lower boiling than acids; many have pleasant, fruity odors.
Low-molecular-weight carboxylic acids and amides are water-soluble; esters are somewhat water-soluble.
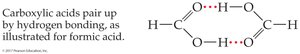
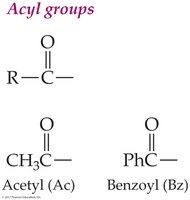
Acyl Groups
The acyl group is the portion of a carboxylic acid that remains after losing the –OH group. Naming conventions include replacing '-ic' with '-oyl' (e.g., acetyl, benzoyl).
Nomenclature of Carboxylic Acids, Esters, and Amides
IUPAC and Common Names
Carboxylic acids: Replace the final '-e' of the alkane with '-oic acid' (e.g., ethanoic acid).
Common names often use Greek letters (α, β, γ) for carbon positions instead of numbers.
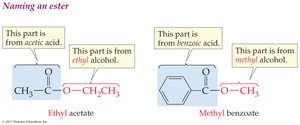
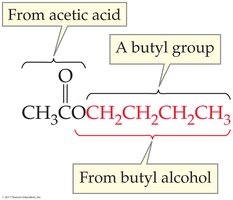
Esters: Name consists of two words—the alkyl group (from alcohol) and the acid part with '-ate' ending.
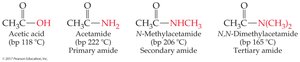
Amides: Replace '-ic acid' or '-oic acid' with '-amide'; alkyl substituents on nitrogen are specified with 'N-'.
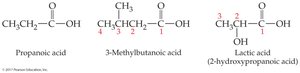

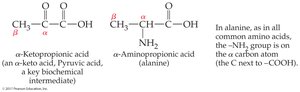


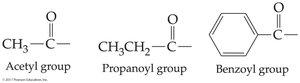
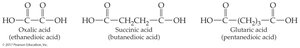
Table: Common Carboxylic Acids
Structure | Common Name |
|---|---|
HCOOH | Formic |
CH3COOH | Acetic |
CH3CH2COOH | Propionic |
CH3(CH2)2COOH | Butyric |
CH3(CH2)4COOH | Valeric |
CH3(CH2)16COOH | Stearic |
HOOC-(CH2)2-COOH | Succinic |
HOOC-(CH2)4-COOH | Glutaric |
H2C=CHCOOH | Acrylic |
COOH-benzene | Benzoic |
COOH-benzene-OH | Salicylic |
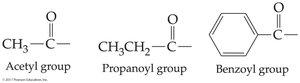

Reactions of Carboxylic Acids and Their Derivatives
Carbonyl-Group Substitution
Carboxylic acids and derivatives undergo substitution reactions where the group attached to the carbonyl carbon is replaced by another group (–Z).
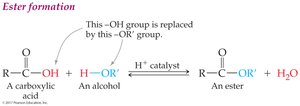
Esterification and Hydrolysis
Esterification: Carboxylic acid reacts with alcohol (in presence of acid catalyst) to form ester and water.
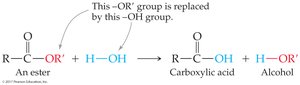
Hydrolysis: Ester reacts with water to yield carboxylic acid and alcohol; reaction is reversible.
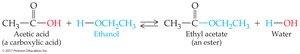
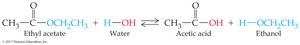
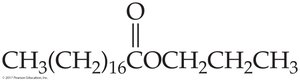
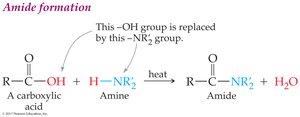
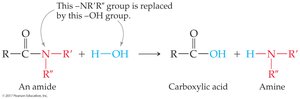
Amide Formation
Carboxylic acids react with ammonia or amines (with heat) to form amides and water.
Primary and secondary amides can hydrogen bond; tertiary amides cannot.

Acidity of Carboxylic Acids
Acid Dissociation and pKa
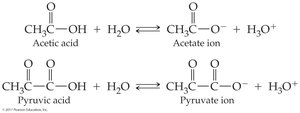
Carboxylic acids are weak acids, establishing equilibrium with carboxylate anions in water.
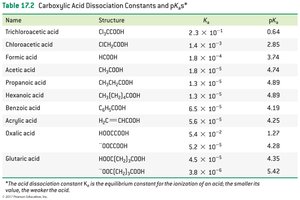
Acidity is measured by acid dissociation constant () and pKa ().
Lower pKa means stronger acid.
Neutralization
Carboxylic acids react with strong bases to form water and carboxylic acid salts.

Effect of Substituents on Acidity
Electronegative substituents (e.g., Cl) increase acidity by stabilizing the carboxylate anion.
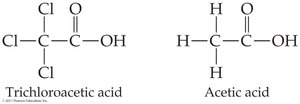
Formation and Hydrolysis of Esters and Amides
Ester and Amide Formation
Esterification: Carboxylic acid + alcohol (acid catalyst) → ester + water
Amide formation: Carboxylic acid + amine (heat) → amide + water
Hydrolysis
Esters and amides can be hydrolyzed to yield carboxylic acids and alcohols/amines.
Acid-catalyzed hydrolysis is the reverse of esterification.
Base-catalyzed hydrolysis of esters is called saponification.

Polyamides and Polyesters
Formation and Uses
Polyamides (e.g., nylon) are formed by reaction of diamines with diacids.
Polyesters (e.g., PET) are formed by reaction of diacids with dialcohols.
These polymers have important industrial and biological applications.
Phosphoric Acid Derivatives
Phosphate Esters and Biological Importance
Phosphoric acid can form mono-, di-, and triesters with alcohols.
Phosphate esters are acidic and exist as ions in neutral or alkaline solutions.
Phosphoryl group transfer (phosphorylation) is a key regulatory mechanism in biochemistry, often involving ATP.

Summary Table: Properties of Carboxylic Acids, Esters, and Amides
Compound | Hydrogen Bonding | Boiling Point | Odor | Acidity | Water Solubility |
|---|---|---|---|---|---|
Carboxylic Acid | Strong | High | Pungent | Acidic | High (small acids) |
Ester | None | Lower | Fruity | Neutral | Moderate |
Amide (primary/secondary) | Strong | High | Odorless | Neutral | High (small amides) |
Amide (tertiary) | None | Lower | Odorless | Neutral | Moderate |
Key Equations
Acid dissociation:
Esterification:
Amide formation:
Hydrolysis of esters:
Hydrolysis of amides:
Additional info:
Carboxylic acids, esters, and amides are central to metabolic pathways and industrial chemistry.
Phosphate esters are crucial in energy transfer and regulation in biological systems.
