 Back
BackAdaptive Immunity: Mechanisms and Components
Study Guide - Smart Notes

Adaptive Immunity
Introduction to Adaptive Immunity
Adaptive immunity refers to the specific defenses that target particular pathogens. It is characterized by a primary response, which is slow and occurs upon first exposure to a foreign substance, and a secondary response, which is faster and stronger due to the presence of memory cells. This system provides the basis for immunity.
Overview of the Adaptive Immune System
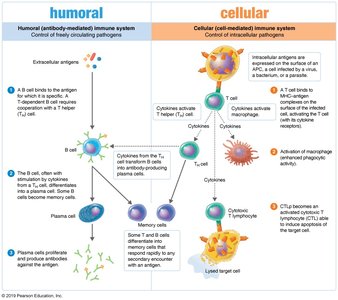
Humoral and Cellular Immunity
Humoral Immunity: Involves B lymphocytes (B cells) that produce antibodies targeting extracellular pathogens such as bacteria, toxins, and viruses.
Cellular (Cell-Mediated) Immunity: Involves T lymphocytes (T cells) that recognize antigens processed by phagocytic cells and are effective against intracellular infections.
Cytokines
Role and Types of Cytokines
Cytokines are chemical messengers produced by immune cells in response to stimuli. They act only on cells with specific receptors and can stimulate the production of more cytokines, sometimes resulting in a positive feedback loop. Overproduction can lead to a cytokine storm, causing tissue damage.
Interleukins: Mediate communication between leukocytes.
Chemokines: Induce migration of leukocytes.
Interferons (IFNs): Interfere with viral infections.
Tumor Necrosis Factor (TNF): Promotes inflammation; excessive amounts are linked to autoimmune diseases.
Superantigens can provoke a strong immune response, leading to toxic shock syndrome, a rare case where the immune reaction damages self-tissues.
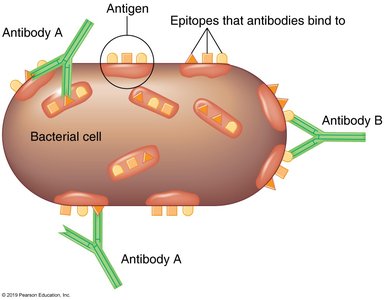
Antigens and Epitopes
Definition and Structure
Antigens are foreign substances that induce the production of antibodies or activate T cells. They are typically components of microbes (e.g., capsules, cell walls, flagella) or foreign molecules like toxins, usually proteins or carbohydrates. Antibodies interact with specific regions on antigens called epitopes (antigenic determinants).
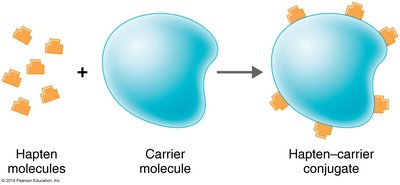
Haptens
Haptens are small molecules that are not immunogenic by themselves but can elicit an immune response when attached to larger carrier molecules.
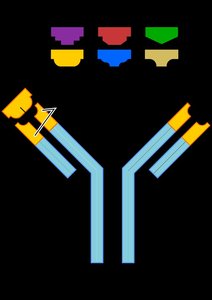
Antibodies (Immunoglobulins)
Structure and Classes
Antibodies, or immunoglobulins (Ig), are globular proteins with a Y-shaped structure composed of two identical light chains and two identical heavy chains joined by disulfide bonds. The variable (V) regions at the tips of the Y arms are the antigen-binding sites, while the constant (C) region forms the stem (Fc region), which can bind to complement proteins. Antibodies can exist as monomers, dimers, or pentamers, with most being monomers.
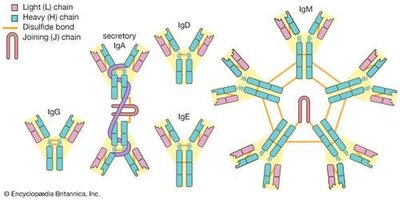
Classes of Immunoglobulins
Class | Structure | % Total Serum Antibody | Location | Functions |
|---|---|---|---|---|
IgG | Monomer | 80% | Blood, lymph, intestine | Enhances phagocytosis, neutralizes toxins/viruses, crosses placenta, long-lived |
IgM | Pentamer | 6% | Blood, lymph, B cell surface (as monomer) | First response to infection, agglutination, complement activation, short-lived |
IgA | Dimer (secretions), monomer (serum) | 13% | Secretions (mucus, saliva, tears, breast milk) | Prevents microbial attachment to mucous membranes |
IgD | Monomer | 0.02% | Blood, lymph, B cell surface | Initiates immune response, may delete self-reactive B cells |
IgE | Monomer | 0.002% | Mast cells, basophils, blood | Allergic reactions, lysis of parasitic worms |

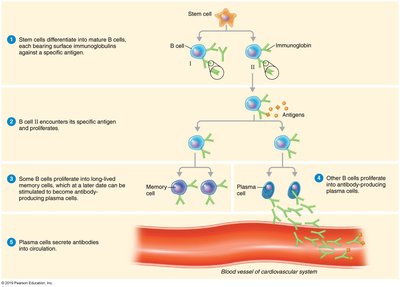
Life Cycle of B Cells
Development and Activation
Production: B cells are produced in the red bone marrow as immature lymphocytes.
Maturation: Self-reactive B cells are destroyed (clonal deletion); mature B cells remain.
Migration: B cells migrate to secondary lymphatic organs (spleen, lymph nodes).
Antigen Encounter: B cells wait to encounter their specific antigen.
Activation: Upon antigen encounter and T cell confirmation, B cells are activated (clonal selection).
Proliferation: Activated B cells proliferate (clonal expansion) into plasma cells and memory cells.
Antibody Production: Plasma cells secrete antibodies; memory cells provide long-term immunity.
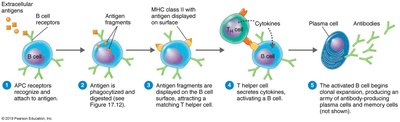
Activation of B Cells
B cells have surface immunoglobulins (Igs) that act as antigen receptors. Upon binding and internalizing an antigen, B cells present antigen fragments on MHC class II molecules. T helper cells recognize these fragments and release cytokines, confirming and activating the B cell, which then proliferates.
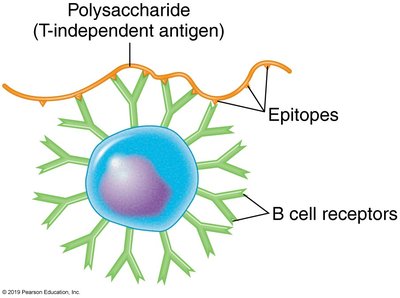
T-Dependent and T-Independent Antigens
T-dependent antigens: Require T helper cells for B cell activation and antibody production.
T-independent antigens: Stimulate B cells without T cell help, usually producing IgM and no memory cells.
Results of Antibody Production
Antigen–Antibody Complexes and Their Effects
When antibodies bind to antigens, they form antigen–antibody complexes that protect the host by tagging foreign molecules or cells for destruction through several mechanisms:
Agglutination: Clumping of antigens for easier removal.
Opsonization: Coating antigens to enhance phagocytosis.
Antibody-dependent cell-mediated cytotoxicity: Targeting large pathogens for destruction by immune cells.
Neutralization: Blocking adhesion of pathogens and toxins.
Activation of complement: Triggering the complement cascade to lyse pathogens.
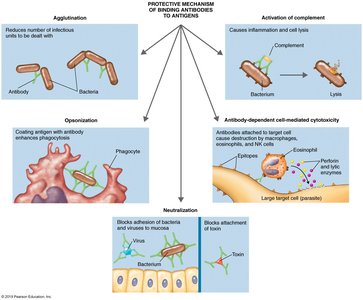
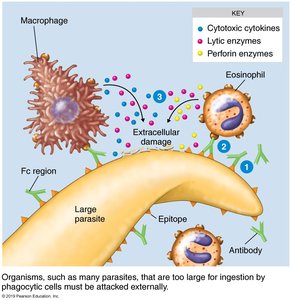
Antibody-Dependent Cell-Mediated Cytotoxicity
Mechanism and Importance
Some pathogens, such as protozoans and helminths, are too large to be phagocytized. Antibodies coat these targets, and immune cells attach to the Fc region of the antibodies, releasing chemicals that lyse the target cell.
Life Cycle of T Cells
Development and Activation
Production: T cells are produced in the red bone marrow.
Maturation: T cells mature in the thymus; self-reactive T cells are destroyed (thymic selection).
Migration: Mature T cells migrate to secondary lymphatic organs.
Antigen Encounter: T cells wait to encounter their specific antigen.
Activation: Requires antigen presentation by APCs and costimulation.
Proliferation: Activated T cells proliferate into effector and memory cells.
Effector Function: Effector T cells carry out immune responses.
Memory: Memory T cells provide long-term immunity.
Activation of T Cells and MHC Molecules
Major histocompatibility complex (MHC) proteins are glycoproteins on cell surfaces. Class I MHC is present on all nucleated cells and marks them as self, while Class II MHC is found only on antigen-presenting cells (APCs) and is crucial for presenting antigens to T cells.
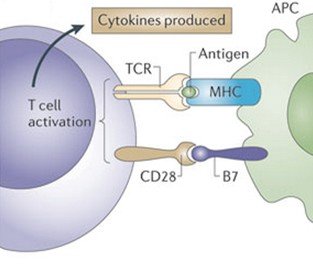
Antigen-Presenting Cells (APCs)
Dendritic cells: Primary APCs that engulf and degrade microbes, presenting antigens with MHC II to T cells.
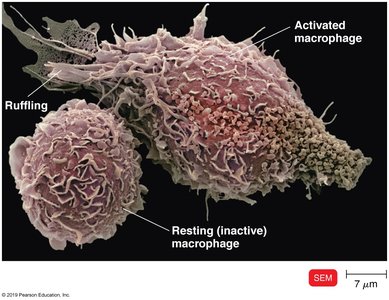
Macrophages: Activated by cytokines or antigen ingestion, migrate to lymph tissue, and present antigens to T cells.
Classes of T Cells
CD4+ and CD8+ T Cells
CD4+ (T helper cells, Th): Bind MHC class II molecules, interact with B cells and APCs, and produce cytokines.
CD8+ (Cytotoxic T lymphocytes, CTL): Bind MHC class I molecules and kill infected self-cells.
T Helper Cells (CD4+)
T helper cells recognize antigen-MHC II complexes on APCs, become activated, and differentiate into subtypes:
TH1: Activate macrophages, enhance complement, stimulate antibody production.
TH2: Activate B cells to produce IgE, activate eosinophils.
TH17: Produce IL-17, contribute to inflammation.
Memory cells: Provide long-term immunity.
T Regulatory Cells (Treg)
T regulatory cells (formerly T suppressor cells) are a subset of CD4+ cells that suppress immune responses against self, protect intestinal bacteria, and help prevent fetal rejection.
T Cytotoxic Cells (CD8+)
Activated cytotoxic T lymphocytes (CTLs) recognize and kill infected self-cells presenting endogenous antigens with MHC class I. CTLs release perforin and enzymes to induce apoptosis (programmed cell death) in the infected cell, preventing the spread of infection.
Extracellular Killing by the Immune System
Natural Killer (NK) Cells
Natural killer cells are lymphocytes that destroy cells lacking MHC class I self-antigens, including virus-infected and tumor cells. They form pores in target cells, leading to lysis or apoptosis, and are less specific than CTLs.
Immunological Memory
Primary and Secondary Responses
The secondary (memory) response is faster, longer-lasting, and stronger than the primary response due to the activation of memory cells. This reflects the intensity of the humoral response and is the basis for effective immunity.
Types of Adaptive Immunity
Active and Passive Immunity
Naturally acquired active immunity: Resulting from infection.
Naturally acquired passive immunity: Transfer of antibodies from mother to child via placenta or milk.
Artificially acquired active immunity: Vaccination (immunization).
Artificially acquired passive immunity: Injection of antibodies, antitoxins, or antivenoms.
Serology and Antiserum
Definitions
Antiserum: Blood-derived fluids containing antibodies, used for passive immunity (e.g., Ebola treatment).
Globulins: Serum proteins, including gamma globulin, which contains all antibody classes.
Serology: The study of reactions between antibodies and antigens.
