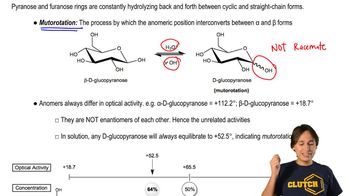
Like glucose, galactose mutarotates when it dissolves in water. The specific rotation of α-D-galactopyranose is +150.7°, and that of the β anomer is +52.8°. When either of the pure anomers dissolves in water, the specific rotation gradually changes to +80.2°. Determine the percentages of the two anomers present at equilibrium.

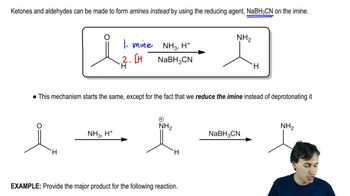
When D-glucose is reduced with sodium borohydride, optically active glucitol results. When optically active D-galactose is reduced, however, the product is optically inactive. Explain this loss of optical activity.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Optical Activity
Chirality and Stereoisomers

Reduction Reactions
Draw and name the products of bromine water oxidation of
(a) D-mannose
(b) D-galactose
(c) D-fructose
The carbonyl group in D-galactose may be isomerized from C1 to C2 by brief treatment with dilute base (by the enediol rearrangement). The product is the C4 epimer of fructose. Draw the furanose structure of the product.
Two sugars, A and B, are known to be glucose and galactose, but it is not certain which one is which. On treatment with nitric acid, A gives an optically inactive aldaric acid, while B gives an optically active aldaric acid. Which sugar is glucose, and which is galactose?
Draw and name the products of nitric acid oxidation of
(a) D-mannose
(b) D-galactose
Ribose, the C2 epimer of arabinose, is most stable in its furanose form. Draw D-ribofuranose.
