Answer the following questions about the eight aldopentoses:
a. Which are enantiomers?
b. Which are C-2 epimers?

 Verified step by step guidance
Verified step by step guidance
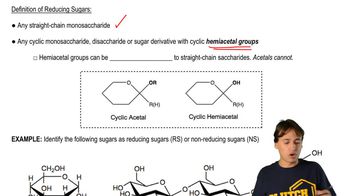
Answer the following questions about the eight aldopentoses:
a. Which are enantiomers?
b. Which are C-2 epimers?
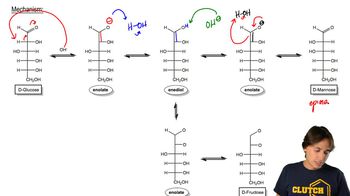
Name the epimers of D-glucose.
Identify the sugar in each description.
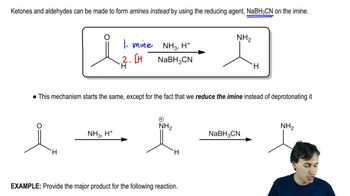
a. An aldopentose that is not D-arabinose forms D-arabinitol when it is reduced with NaBH4.
What is the configuration of each of the asymmetric centers in the Fischer projection of
a. D-glucose?
Identify the sugar in each description.
b. A sugar that is not D-altrose forms D-altraric acid when it is oxidized with nitric acid.
D-Xylose and D-lyxose are formed when d-threose undergoes a Kiliani–Fischer synthesis. D-Xylose is oxidized to an optically inactive aldaric acid, whereas D-lyxose forms an optically active aldaric acid. What are the structures of D-xylose and D-lyxose?