Textbook Question
a. What percentage of the a-amino group of lysine will be protonated at its pI?
<25%, 50%, >75%
b. Answer the same question for the e-amino group of lysine
1549
views

 Verified step by step guidance
Verified step by step guidance
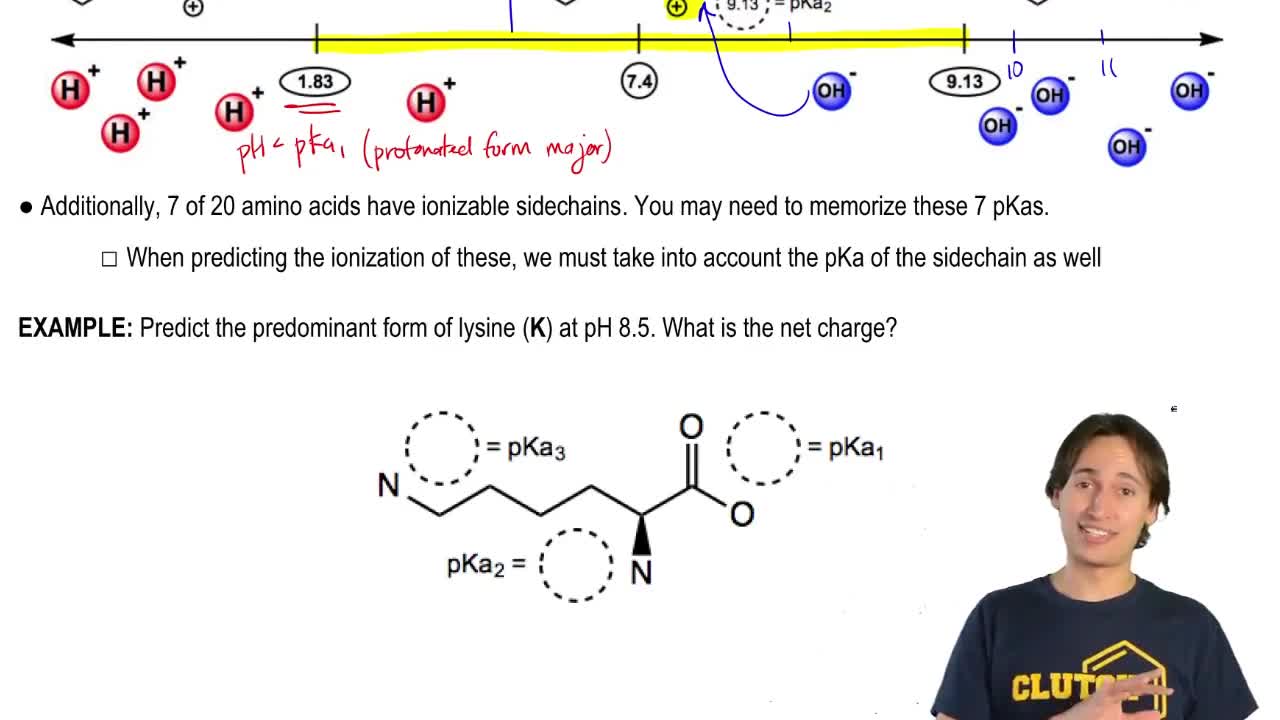

a. What percentage of the a-amino group of lysine will be protonated at its pI?
<25%, 50%, >75%
b. Answer the same question for the e-amino group of lysine
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50:
a. aspartate before serine
Why is excess ammonia used in the preceding reaction?
d. Which amino acid has a greater negative charge at pH = 6.20, glycine or methionine?
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50:
b. serine before alanine
Explain why the pI of lysine is the average of the pKa values of its two protonated amino groups.