Textbook Question
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
1398
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 36d,e,f
Problem 36d,e,f Verified step by step guidance
Verified step by step guidance

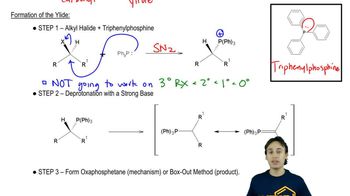
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
a. What alkene is required to synthesize each of the following compounds?
b. What other epoxide is formed in each synthesis?
c. Assign an R or S configuration to each asymmetric center.
2.
What characteristics must the reactant of a stereospecific reaction have?
The following product was obtained from the ozonolysis of an alkene followed by treatment with dimethyl sulfide. What is the structure of the alkene?
c.
What stereoisomers are obtained from each of the following reactions?
b.
a. What stereoisomers are formed in the following reaction? Are they enantiomers or diastereomers?
b. Which stereoisomer is formed in greater yield?