Imagine an electrophilic addition where the first step is exothermic. Which carbocation—2°, 3°, or neither—would you expect to form preferentially? Explain.

Draw a mechanism for the acid-catalyzed conversion of DPP to IPP. How do you know that your mechanism is correct?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Acid-Catalyzed Reactions
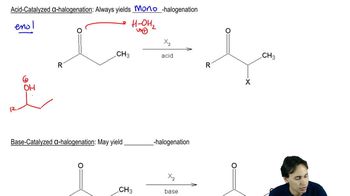
Reaction Mechanism

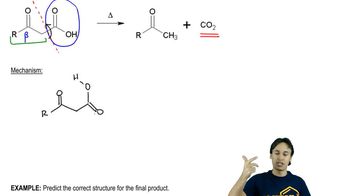
Verification of Mechanisms
What is the expected product for the reaction of each of the following alkenes with (i) HBr and (ii) HCl?
(e)
Provide an arrow-pushing mechanism that rationalizes the formation of each of the products you predicted in Assessment 8.31. Make sure your mechanism accounts for all products formed, including stereoisomers and regioisomers, where applicable.
What is the expected product for the reaction of each of the following alkenes with (i) HBr and (ii) HCl?
(d)
For the enzyme isopentenyl pyrophosphate isomerase, IPP binds tightly as a result of interactions between the active site amino acid residues and the diphosphate of IPP. Without concerning yourself with the structure of amino acids, what charges might you expect to be present in the active site to hold IPP in place so that the enzymatic reactions can occur?
Which would you expect to be more selective for carbocation formation, the electrophilic addition of HF or HBr to 2-methylbut-2-ene? Explain your answer.
