Show the product of the Diels–Alder dimerization of cyclobutadiene. (This reaction is similar to the dimerization of cyclopentadiene, discussed in Section 15-11.)
Ch. 16 - Aromatic Compounds

Chapter 16, Problem 6
Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, and estimate the angle between the p orbitals of adjacent pi bonds.
 Verified step by step guidance
Verified step by step guidance1
Step 1: Cyclooctatetraene is an eight-membered ring with alternating double bonds. In its tub conformation, the ring adopts a non-planar shape to minimize angle strain and steric hindrance.
Step 2: To draw the tub conformation, visualize the ring as having two opposite sides bent upward and downward, forming a shape similar to a bathtub. This conformation allows the molecule to relieve strain caused by the bond angles deviating from the ideal 120° of sp2 hybridized carbons.
Step 3: The p orbitals of adjacent pi bonds in cyclooctatetraene are not perfectly aligned due to the non-planar geometry. This misalignment reduces conjugation and makes the molecule non-aromatic.
Step 4: Estimate the angle between the p orbitals of adjacent pi bonds by considering the geometry of the tub conformation. The angle is typically less than 90° but greater than 0°, as the p orbitals are tilted relative to each other due to the bent structure.
Step 5: Use a molecular model kit or software to construct the tub conformation of cyclooctatetraene. This will help visualize the spatial arrangement of the p orbitals and confirm the estimated angle between them.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
6mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Cyclooctatetraene Structure
Cyclooctatetraene (COT) is an eight-membered cyclic hydrocarbon with alternating double bonds. Its unique structure allows it to adopt various conformations, with the 'tub' conformation being one of the most stable. This conformation minimizes steric strain and allows for a more favorable arrangement of the π bonds, which is crucial for understanding its reactivity and properties.
Recommended video:
Guided course

Drawing Resonance Structures
Conformation and Angle Between p Orbitals
In organic chemistry, conformation refers to the spatial arrangement of atoms in a molecule that can change due to rotation around single bonds. In the case of cyclooctatetraene, the angle between the p orbitals of adjacent π bonds is significant for determining the molecule's reactivity and stability. Estimating this angle helps in understanding the overlap of p orbitals and the resulting π bond character.
Recommended video:
Guided course
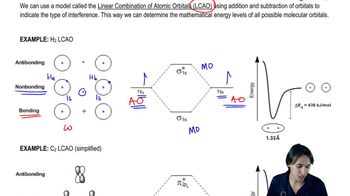
What’s the difference between atomic and molecular orbitals?
Aromaticity and Its Criteria
Aromaticity is a property of cyclic compounds that exhibit enhanced stability due to resonance and delocalization of π electrons. For a compound to be aromatic, it must be cyclic, planar, and follow Hückel's rule, which states that it must have (4n + 2) π electrons. Cyclooctatetraene is not aromatic in its tub conformation, as it does not meet these criteria, which is essential for understanding its chemical behavior.
Recommended video:
Guided course

Intro to Aromaticity
Related Practice
Textbook Question
1054
views
Textbook Question
Classify the following compounds as aromatic, antiaromatic, or nonaromatic.
(c)
(d)
1726
views
Textbook Question
a. Draw the resonance forms of benzene, cyclobutadiene, and cyclooctatetraene, showing all the carbon and hydrogen atoms.
b. Assuming that these molecules are all planar, show how the p orbitals on the sp2 hybrid carbon atoms form continuous rings of overlapping orbitals above and below the plane of the carbon atoms.
1002
views
Textbook Question
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic.
896
views
Textbook Question
Classify the following compounds as aromatic, antiaromatic, or nonaromatic.
(a)
(b)
1090
views
Textbook Question
Does the MO energy diagram of cyclooctatetraene (Figure 16-8) appear to be a particularly stable or unstable configuration? Explain.
1970
views
1
rank
