Predict the products of the reactions of the following compounds with:
1. chromic acid or excess sodium hypochlorite with acetic acid.
2. PCC or NaOCl (1 equivalent) with TEMPO.
d. cyclohexanone



 Verified step by step guidance
Verified step by step guidance


Predict the products of the reactions of the following compounds with:
1. chromic acid or excess sodium hypochlorite with acetic acid.
2. PCC or NaOCl (1 equivalent) with TEMPO.
d. cyclohexanone
Predict the products of the reactions of the following compounds with:
1. chromic acid or excess sodium hypochlorite with acetic acid.
2. PCC or NaOCl (1 equivalent) with TEMPO.
e. cyclohexane
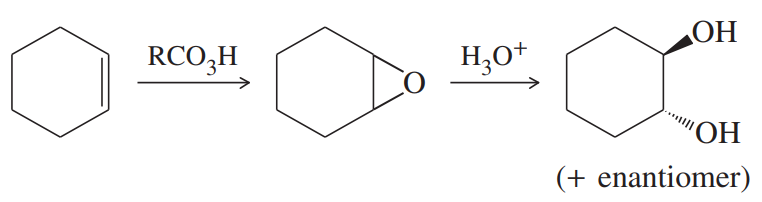
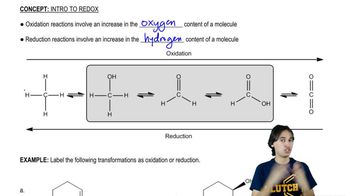
Classify each reaction as an oxidation, a reduction, or neither.
(g)
(h)
(i)
Predict the products of the reactions of the following compounds with:
1. chromic acid or excess sodium hypochlorite with acetic acid.
2. PCC or NaOCl (1 equivalent) with TEMPO.
a. cyclohexanol
b. 1-methylcyclohexanol
c. cyclopentylmethanol
Classify each reaction as an oxidation, a reduction, or neither.
(d)
(e)
(f)
Classify each reaction as an oxidation, a reduction, or neither.
(a)
(b)
(c)