Textbook Question
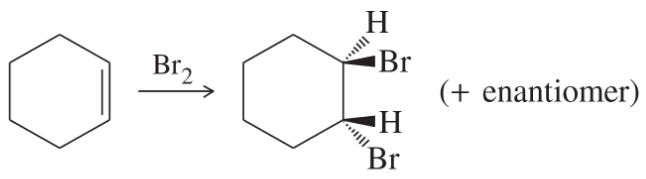
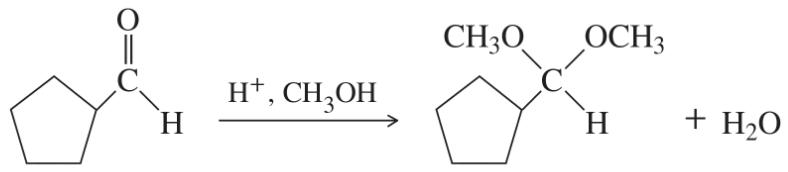
Classify each reaction as an oxidation, a reduction, or neither.
(j)
(k)
(l)
949
views


 Verified step by step guidance
Verified step by step guidance



Classify each reaction as an oxidation, a reduction, or neither.
(j)
(k)
(l)
Classify each reaction as an oxidation, a reduction, or neither.
(g)
(h)
(i)
Predict the products of the reactions of the following compounds with:
1. chromic acid or excess sodium hypochlorite with acetic acid.
2. PCC or NaOCl (1 equivalent) with TEMPO.
a. cyclohexanol
b. 1-methylcyclohexanol
c. cyclopentylmethanol
Classify each reaction as an oxidation, a reduction, or neither.
(a)
(b)
(c)