How would you make the following compounds from N-benzylbenzamide?
a. dibenzylamine

 Bruice 8th Edition
Bruice 8th Edition Ch. 16 - Reactions of Aldehydes and Ketones • More Reactions of Carboxylic Acid Derivatives
Ch. 16 - Reactions of Aldehydes and Ketones • More Reactions of Carboxylic Acid Derivatives Problem 24
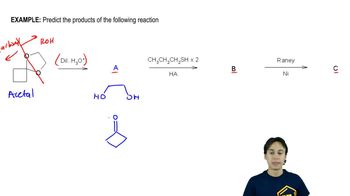
Problem 24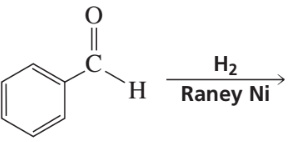
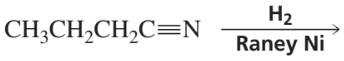
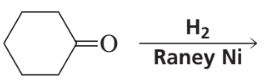
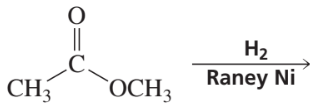
 Verified step by step guidance
Verified step by step guidance


How would you make the following compounds from N-benzylbenzamide?
a. dibenzylamine
Why is the pKa value of protonated hydroxylamine (6.0) so much lower than the value of a protonated primary amine such as protonated methylamine (10.7)?
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate formed in this reaction, and show how it can be converted to a ketone.
How would you make the following compounds from N-benzylbenzamide?
c. benzyl alcohol
The pKa of protonated acetone is about –7.5 and the pKa of protonated hydroxylamine is 6.0.
a. In a reaction with hydroxylamine at pH 4.5 (Figure 16.2), what fraction of acetone is present in its acidic, protonated form?
<IMAGE>
How would you make the following compounds from N-benzylbenzamide?
b. benzoic acid