Textbook Question
The Gabriel synthesis is most frequently done with 1° alkyl halides. Why is it less successful with more substituted halides?
1057
views


 Verified step by step guidance
Verified step by step guidance

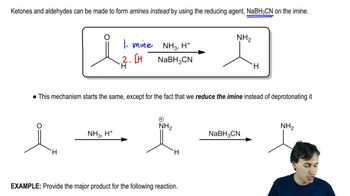

The Gabriel synthesis is most frequently done with 1° alkyl halides. Why is it less successful with more substituted halides?
Predict the product(s) of the reactions shown.
(b)
The synthesis of ⍺-hydroxy acids can be done starting with amino acids. Suggest a mechanism of the two-step transformation shown. [The alcohol oxygen is the same in the reactant and product.
Predict the product(s) of the reactions shown.
(j)
The final step in the synthesis of diazoxide, a drug used to treat low blood pressure, is shown here. Suggest a mechanism for this step.
Predict the product(s) of the reactions shown.
(i)