Predict the product of the following reactions.
(c)


 Verified step by step guidance
Verified step by step guidance
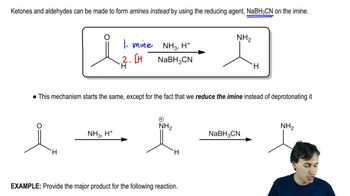


Predict the product of the following reactions.
(c)
We usually calculate Keq for acid–base reactions using pKa values.
(a) Derive an equation to calculate Keq using pKb values, then
(b) use it to calculate the equilibrium constant for the following reaction.
Which would you expect to be a stronger nucleophile, ethyl amine or diethyl amine? Why?
The nitro group directs electrophilic aromatic substitution to the meta position. After reduction by hydrogenation, to which carbon(s) does it direct?
Given the pKb, calculate the pKa of the conjugate acid.
(b)
Following a LiAlH₄ reduction, an IR spectrum suggested that the product was a mixture of the starting amide and the desired amine.
(b) Once separated, how could you distinguish between the amide and the amine using IR spectroscopy?