Textbook Question
What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?
b.
1467
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 95a
Problem 95a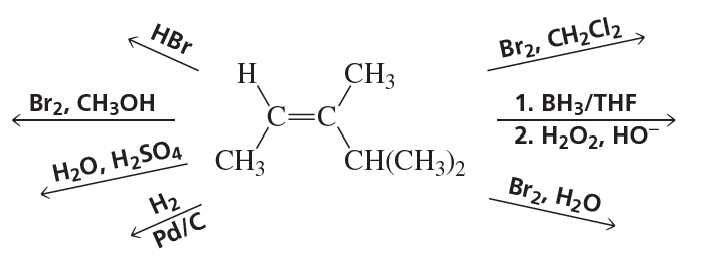
 Verified step by step guidance
Verified step by step guidance


What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?
b.
a. Propose a mechanism for the following reaction:
b. Is the initially formed carbocation primary, secondary, or tertiary?
c. Is the rearranged carbocation primary, secondary, or tertiary?
d. Why does the rearrangement occur?
Draw the products of the following reactions, including their configurations:
Draw the products of the following reactions, including their configurations:
What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?
a.
What alkene gives the product shown after reaction first with ozone and then with dimethyl sulfide?
c.