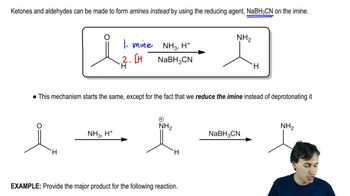
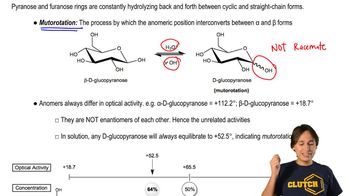
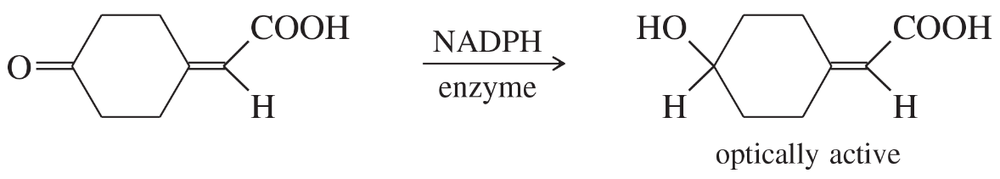The original definition of meso is 'an achiral compound that has chiral diastereomers.' Our working definition of meso is 'an achiral compound that has chiral centers (usually asymmetric carbon atoms).' The working definition is much easier to apply, because we don't have to envision all possible chiral diastereomers of the compound. Still, the working definition is not quite as complete as the original definition.
a. Show how cis-cyclooctene is defined as a meso compound under the original definition, but not under our working definition. (Review Figure 5-19)