Chlorine exists as one of two isotopes with atomic masses of 34.969 amu (35Cl) and 36.966 amu (37Cl) . Calculate the relative abundance of 35Cl and 37Cl based on the average atomic mass of 35.453 amu.
Ch. 2 - General Chemistry Translated: Finding the Electrons

Mullins1st EditionOrganic Chemistry: A Learner Centered ApproachISBN: 9780137566471Not the one you use?Change textbook
All textbooks Mullins 1st Edition
Mullins 1st Edition Ch. 2 - General Chemistry Translated: Finding the Electrons
Ch. 2 - General Chemistry Translated: Finding the Electrons Problem 5
Problem 5
 Mullins 1st Edition
Mullins 1st Edition Ch. 2 - General Chemistry Translated: Finding the Electrons
Ch. 2 - General Chemistry Translated: Finding the Electrons Problem 5
Problem 5Chapter 1, Problem 5
Why is argon considered to be so stable that it is referred to as a noble gas?
 Verified step by step guidance
Verified step by step guidance1
Argon is considered stable because it has a complete octet in its outermost electron shell, meaning it has 8 valence electrons. This configuration is energetically favorable and does not require argon to gain, lose, or share electrons.
The term 'noble gas' refers to elements in Group 18 of the periodic table, which are characterized by their full valence electron shells. Argon, being part of this group, shares this property.
Due to its full valence shell, argon has minimal chemical reactivity. It does not readily form bonds with other elements, making it chemically inert under standard conditions.
The stability of argon is further supported by its electronic configuration: \( 1s^2 2s^2 2p^6 3s^2 3p^6 \). This configuration reflects a completely filled outer shell, which is a hallmark of noble gases.
In practical applications, argon's inertness is utilized in environments where chemical reactions need to be minimized, such as in welding or as a protective atmosphere for sensitive materials.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Was this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Noble Gases
Noble gases are a group of chemical elements in Group 18 of the periodic table, characterized by their lack of reactivity due to having a complete valence shell of electrons. This stable electron configuration makes them unlikely to form chemical bonds with other elements, which is why they are often referred to as 'inert' or 'noble.'
Recommended video:
Guided course
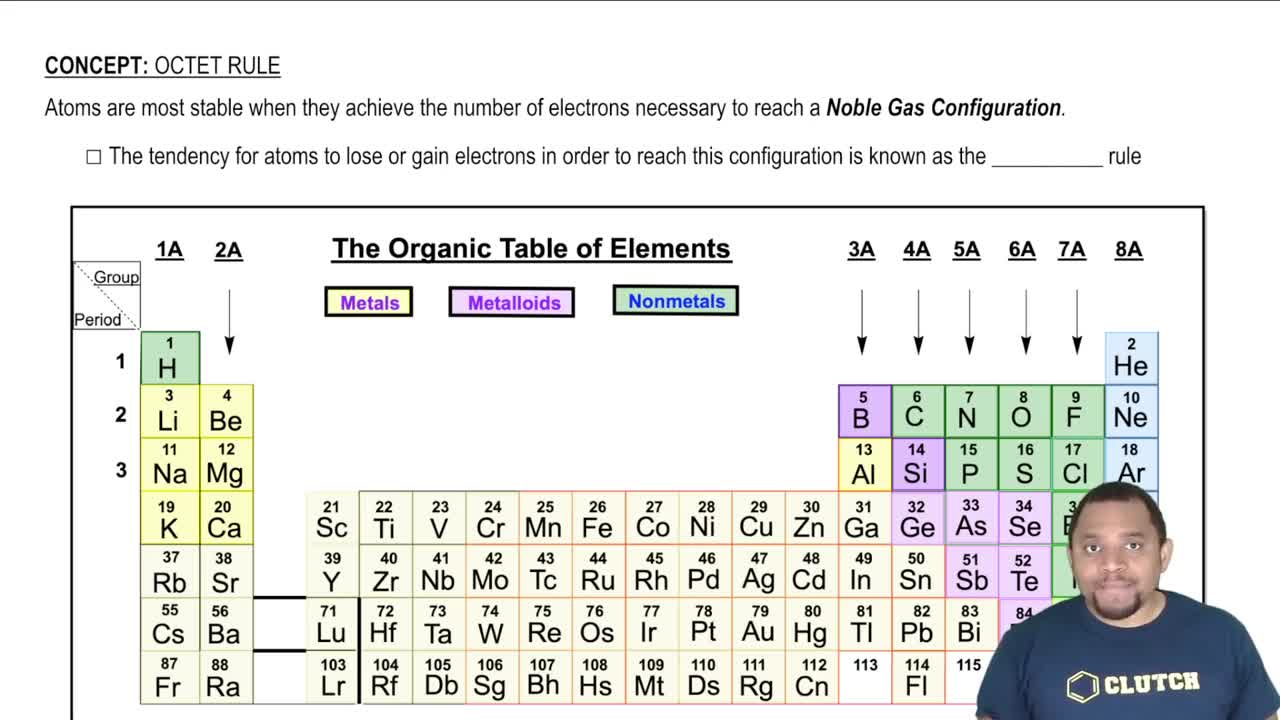
How Noble gases are related to the octet rule.
Electron Configuration
Electron configuration describes the distribution of electrons in an atom's orbitals. For argon, the electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶, indicating that it has eight electrons in its outer shell, fulfilling the octet rule. This full outer shell is a key reason for its stability and lack of reactivity.
Recommended video:
Guided course

The Electron Configuration
Reactivity of Elements
The reactivity of elements is influenced by their electron configurations, particularly the number of electrons in their outermost shell. Elements with incomplete outer shells tend to be more reactive as they seek to gain, lose, or share electrons to achieve stability. In contrast, argon’s complete outer shell results in minimal tendency to react with other elements.
Recommended video:
Guided course

Understanding the Element Effect.
Related Practice
Textbook Question
1336
views
Textbook Question
A chemistry student drew the following incorrect electron configuration for carbon. (a) Correct the diagram.
861
views
Textbook Question
How many valence shell electrons do each of the following elements contain? How many new bonds can each form?
(c) O
1283
views
Textbook Question
How many valence shell electrons do each of the following elements contain? How many new bonds can each form?
(a) C
1265
views
Textbook Question
How many valence shell electrons do each of the following elements contain? How many new bonds can each form?
(b) N
1808
views
Textbook Question
Bromine-79 (50.7% abundance) has an atomic mass of 78.918 amu, whereas bromine-81 (49.3% abundance) has an atomic mass of 80.916 amu. From these data, calculate the average atomic mass of bromine that you would expect to see in the periodic table.
1281
views