How might you make the catalytic cycle in Figure 9.31 more sustainable while still using NMO as the co-oxidant?
<IMAGE>

 Verified step by step guidance
Verified step by step guidance



How might you make the catalytic cycle in Figure 9.31 more sustainable while still using NMO as the co-oxidant?
<IMAGE>
Which of molecules A–D would you expect to give a positive permanganate test? That is, which would result in a purple KMnO₄ solution turning brown?
(c)
Which of molecules A–D would you expect to give a positive permanganate test? That is, which would result in a purple KMnO₄ solution turning brown?
(d)
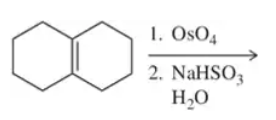
When using a terminal alkene under the conditions shown here, explain why it is unnecessary to show the relative stereochemical outcome in the product.
Predict the product(s) of each of the following reactions, making sure to indicate the relative stereochemical outcome. Indicate any racemic mixtures by drawing both enantiomers.
(b)
When producing a chiral molecule, epoxide formation still results in a mixture of enantiomers, despite its stereospecificity.
(b) How is it that a reaction can be stereospecific while still producing two enantiomers?