Cellulose is converted to cellulose acetate by treatment with acetic anhydride and pyridine. Cellulose acetate is soluble in common organic solvents, and it is easily dissolved and spun into fibers. Show the structure of cellulose acetate.

Cytosine, uracil, and guanine have tautomeric forms with aromatic hydroxy groups. Draw these tautomeric forms.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Tautomerism
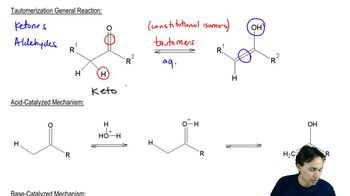
Aromatic Hydroxy Groups

Nucleobase Structure

An aliphatic aminoglycoside is relatively stable to base, but it is quickly hydrolyzed by dilute acid. Propose a mechanism for the acid-catalyzed hydrolysis.
Ribonucleosides are not so easily hydrolyzed, requiring relatively strong acid. Using your mechanism for part (a), show why cytidine and adenosine (for example) are not so readily hydrolyzed. Explain why this stability is important for living organisms.
Trehalose is a nonreducing disaccharide (C12H22O11) isolated from the poisonous mushroom Amanita muscaria. Treatment with an α-glucosidase converts trehalose to two molecules of glucose, but no reaction occurs when trehalose is treated with a β-glucosidase. When trehalose is methylated by dimethyl sulfate in mild base and then hydrolyzed, the only product is 2,3,4,6-tetra-O-methylglucose. Propose a complete structure and systematic name for trehalose.
Raffinose is a trisaccharide (C18H32O16) isolated from cottonseed meal. Raffinose does not reduce Tollens reagent, and it does not mutarotate. Complete hydrolysis of raffinose gives D-glucose, D-fructose, and D-galactose. When raffinose is treated with invertase, the products are D-fructose and a reducing disaccharide called melibiose. Raffinose is unaffected by treatment with a β-galactosidase, but an α-galactosidase hydrolyzes it to D-galactose and sucrose. When raffinose is treated with dimethyl sulfate and base followed by hydrolysis, the products are 2,3,4-tri-O-methylglucose, 1,3,4,6-tetra-O-methylfructose, and 2,3,4,6-tetra-O-methylgalactose. Determine the complete structures of raffinose and melibiose, and give a systematic name for melibiose.
All of the rings of the four heterocyclic bases are aromatic. This is more apparent when the polar resonance forms of the amide groups are drawn, as is done for thymine here. Redraw the hydrogen-bonded guanine-cytosine and adenine-thymine pairs shown in Figure 23-24, using the polar resonance forms of the amides. Show how these forms help to explain why the hydrogen bonds involved in these pairings are particularly strong. Remember that a hydrogen bond arises between an electron-deficient hydrogen atom and an electron-rich pair of nonbonding electrons.
