Show each step in the mechanism of the acid-catalyzed interconversion of (R)- and (S)-3-methylpentan-2-one.
Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds

All textbooks Wade 9th Edition
Wade 9th Edition Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds
Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds Problem 1c3
Problem 1c3
 Wade 9th Edition
Wade 9th Edition Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds
Ch. 22 - Condensations and Alpha Substitutions of Carbonyl Compounds Problem 1c3
Problem 1c3Chapter 22, Problem 1c3
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the first enol in base.
 Verified step by step guidance
Verified step by step guidance1
Identify the structure of phenylacetone: It is a ketone with a phenyl group attached to the alpha-carbon. The molecular structure can be represented as C6H5-CH2-CO-CH3.
Understand the concept of enol formation: Enols are formed when a hydrogen atom from the alpha-carbon (the carbon adjacent to the carbonyl group) is removed, and the resulting negative charge is delocalized to form a double bond between the alpha-carbon and the carbonyl carbon, while the oxygen of the carbonyl group gains a proton to form an -OH group.
Initiate the mechanism in base: A strong base (e.g., hydroxide ion, OH⁻) abstracts an acidic proton from the alpha-carbon of phenylacetone. This generates a resonance-stabilized enolate ion.
Illustrate resonance stabilization: The enolate ion has two resonance forms. In one form, the negative charge is on the alpha-carbon, and in the other, the negative charge is delocalized onto the oxygen atom of the carbonyl group. This delocalization stabilizes the intermediate.
Complete the enol formation: The enolate ion reacts with a proton donor (e.g., water) to protonate the oxygen atom, forming the enol. The final structure of the enol will have a double bond between the alpha-carbon and the carbonyl carbon, and the oxygen will be part of a hydroxyl (-OH) group.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
2mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Enol Formation
Enol formation involves the conversion of a carbonyl compound, such as a ketone or aldehyde, into its corresponding enol form, which features a double bond between a carbon and an adjacent carbon with a hydroxyl group. This process is significant in organic chemistry as enols can participate in various reactions, including tautomerization and electrophilic addition.
Recommended video:
Guided course

Formation of Enolates
Base-Catalyzed Mechanism
In a base-catalyzed mechanism, a base abstracts a proton from the carbon adjacent to the carbonyl group, generating a carbanion. This carbanion can then attack the carbonyl carbon, leading to the formation of the enolate ion, which can subsequently rearrange to form the enol. Understanding this mechanism is crucial for predicting the products of reactions involving carbonyl compounds.
Recommended video:
Guided course
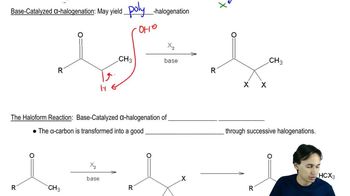
Base Catalyzed
Tautomerization
Tautomerization is a chemical reaction that involves the interconversion between two isomers, typically a keto form and an enol form. This process is often facilitated by the presence of acids or bases and is important in understanding the stability and reactivity of different isomers in organic compounds, including phenylacetone.
Recommended video:
Guided course
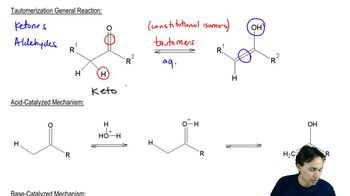
Tautomerization Mechanisms
Related Practice
Textbook Question
1492
views
Textbook Question
When cis-2,4-dimethylcyclohexanone is dissolved in aqueous ethanol containing a trace of NaOH, a mixture of cis and trans isomers results. Propose a mechanism for this isomerization.
1298
views
Textbook Question
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the second enol in base.
581
views
Textbook Question
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the second enol in acid.
567
views
Textbook Question
Phenylacetone can form two different enols.
(a) Show the structures of these enols.
(b) Predict which enol will be present in the larger concentration at equilibrium.
940
views
Textbook Question
Phenylacetone can form two different enols.
(c) Propose mechanisms for the formation of the first enol in acid.
887
views