Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
c. 1-phenylpropan-2-ol

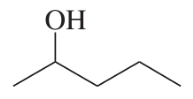
 Verified step by step guidance
Verified step by step guidance



Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
c. 1-phenylpropan-2-ol
Show how you would use Grignard syntheses to prepare the following alcohol from the indicated starting material and any other necessary reagents.
(a) octan-3-ol from hexanal, CH3(CH2)4CHO
Show how you would synthesize the following alcohol from appropriate alkene.
(d)
Show how you would synthesize the following alcohol from appropriate alkene.
(b)
Write structures for a homologous series of alcohols (R―OH) having from one to six carbons.
Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
d. 3-phenylprop-2-en-1-ol