Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
c. 1-phenylpropan-2-ol

 Verified step by step guidance
Verified step by step guidance

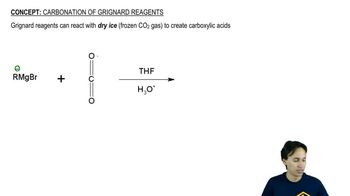

Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
c. 1-phenylpropan-2-ol
Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
b. 1-phenylpropene
Draw the organic products you would expect to isolate from the following reactions (after hydrolysis).
(m)
Draw the organic products you would expect to isolate from the following reactions (after hydrolysis).
(p)
Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent parts of this problem.
d. 3-phenylprop-2-en-1-ol
Draw the organic products you would expect to isolate from the following reactions (after hydrolysis).
(n)