Which of the following are optically active?

Draw the structure for a compound with molecular formula C2H2I2F2
a. that is optically inactive because it does not have an asymmetric center.
b. that is optically inactive because it is a meso compound.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
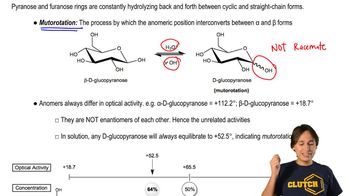
Key Concepts
Optical Activity
Chirality and Asymmetric Centers

Meso Compounds

Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
e.
f.
Assign relative priorities to each set of substituents:
c. -C(=O)CH3, -CH=CH2, -Cl, -C=N
Draw the structure for a compound with molecular formula C2H2I2F2
c. that is optically active.
For many centuries, the Chinese have used extracts from a group of herbs known as ephedra to treat asthma. A compound named ephedrine has been isolated from these herbs and found to be a potent dilator of air passages in the lungs.
a. How many stereoisomers does ephedrine have?
b. The stereoisomer shown here is the one that is pharmacologically active. What is the configuration of each of the asymmetric centers?
Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
c.
d.
