For the molecules shown,
(i) count the number of stereocenters present and
(ii) draw all possible stereoisomers.
(iii) Identify the relationships between stereoisomers as enantiomers or diastereomers.
(b)

 Verified step by step guidance
Verified step by step guidance


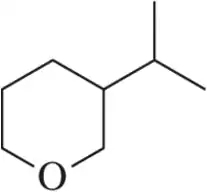
For the molecules shown,
(i) count the number of stereocenters present and
(ii) draw all possible stereoisomers.
(iii) Identify the relationships between stereoisomers as enantiomers or diastereomers.
(b)
Imagine a sample that is enriched in the R enantiomer. If the % ee of the sample is 83%,
(a) what percent of the mixture is racemic?
(b) What is the ratio of R to S?
How many stereoisomers are possible for each of the following molecules?
(a)
(S)-Propranolol, a drug used for the treatment of anxiety, has a specific rotation of -25.5° . Attempting to prepare it in enantiopure form, a chemist produced a compound that gave a specific rotation of -18.3° . What is the ratio of (S)- to (R)-propranolol produced by the chemist?
(R)-Selegiline, a monoamine oxidase (MAO) inactivator, was approved by the FDA in 1989 for the treatment of Parkinson's disease. In pure form, it has a specific rotation, [α]²⁰D = - 11.0°. What is the expected specific rotation of a mixture containing 64% S and 36% R?
For the molecules shown,
(i) count the number of stereocenters present and
(ii) draw all possible stereoisomers.
(iii) Identify the relationships between stereoisomers as enantiomers or diastereomers.
(c)