Textbook Question
Predict the product(s) of the following nucleophilic aromatic substitution reactions occurring by the benzyne mechanism.
(b)
456
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions
Ch. 23 - Benzene I: Aromatic Stability and Substitution Reactions Problem 57a
Problem 57a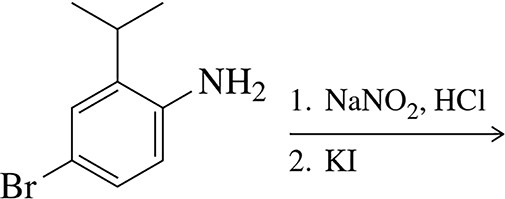
 Verified step by step guidance
Verified step by step guidance

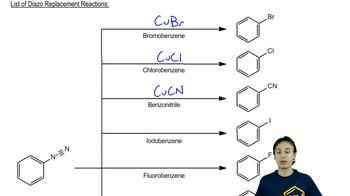

Predict the product(s) of the following nucleophilic aromatic substitution reactions occurring by the benzyne mechanism.
(b)
Predict the products of the following nucleophilic aromatic substitution reactions.
(c)
Predict the products of the following reactions.
(a)
Conversion of t-butylbenzene to a phenol results in two products when the reaction occurs through the benzyne mechanism. Devise a sequence involving a diazonium ion in which only one phenol product is produced.
Assume that Ha is the proton removed in the first step of the benzyne mechanism with p-chlorotoluene. Draw the benzyne intermediate and identify the two products that will result.
Predict the products of the following reactions.
(b)