Textbook Question
Predict the products of the following reactions:
(h)
809
views

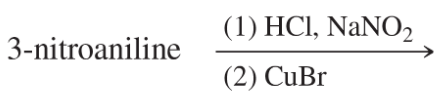
 Verified step by step guidance
Verified step by step guidance
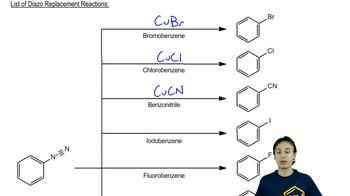


Predict the products of the following reactions:
(h)
Predict the products of the following reactions:
(k)
Predict the products of the following reactions:
(d)
Predict the products of the following reactions:
(g)
Predict the products of the following reactions:
(g)
Predict the products of the following reactions:
(i)