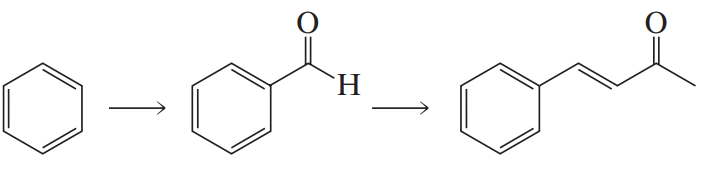
Textbook Question
Identify A–J:
1061
views

 Verified step by step guidance
Verified step by step guidance



Identify A–J:
Identify A–J:
Describe how naphthalene can be prepared from the given starting material.
What reagents are required to carry out the following synthesis?
Describe two synthetic routes for the preparation of p-methoxyaniline from benzene.
Using resonance contributors for the carbocation intermediate, explain why a phenyl group is an ortho–para director.