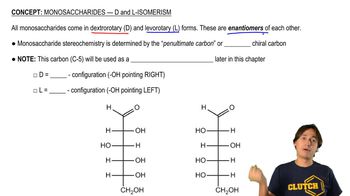
Textbook Question
For each of the following compounds, draw the possible geometric isomers and name each isomer:
a. 2-methyl-2,4-hexadiene
1163
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics
Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics Problem 53b
Problem 53b Verified step by step guidance
Verified step by step guidance


For each of the following compounds, draw the possible geometric isomers and name each isomer:
a. 2-methyl-2,4-hexadiene
Which bond is stronger? Briefly explain why.
a.
How many of the following names are correct? Correct the incorrect names.
a. 3-pentene
b. 2-octene
c. 2-vinylpentane
For each of the following compounds, draw the possible geometric isomers and name each isomer:
d. 3-methyl-2,4-hexadiene
Which bond is stronger? Briefly explain why.
b.
For each of the following compounds, draw the possible geometric isomers and name each isomer:
c. 1,4-pentadiene