Textbook Question
Name the following:
b.
1042
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 7 - The Reactions of Alkynes • An Introduction to Multistep Synthesis
Ch. 7 - The Reactions of Alkynes • An Introduction to Multistep Synthesis Problem 7a
Problem 7a
 Verified step by step guidance
Verified step by step guidance


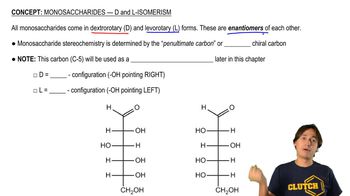
Name the following:
b.
What orbitals are used to form the carbon–carbon s bond between the highlighted carbons?
g.
h.
i.
Draw the condensed and skeletal structures for each of the following:
f. dimethylacetylene
Draw the condensed and skeletal structures for each of the following:
e. 4,4-dimethyl-1-pentyne
Draw the structures and give the common and systematic names for the seven alkynes with molecular formula C6H10.
What orbitals are used to form the carbon–carbon s bond between the highlighted carbons?
a.
b.
c.