Textbook Question
Draw the structures of the compound methyl α-D-galactopyranoside.
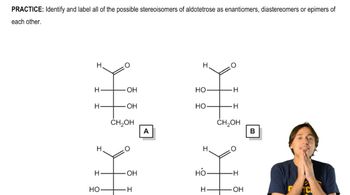
Allose is the C3 epimer of glucose, and ribose is the C2 epimer of arabinose.
822
views

 Verified step by step guidance
Verified step by step guidance

Draw the structures of the compound methyl α-D-galactopyranoside.
Allose is the C3 epimer of glucose, and ribose is the C2 epimer of arabinose.
The mechanism of glycoside formation is the same as the second part of the mechanism for acetal formation. Propose a mechanism for the formation of methyl β-D-glucopyranoside.
Which of the following are reducing sugars? Comment on the common name sucrose for table sugar.
(e)
(f)
Draw the structures of the compound ethyl β-D-ribofuranoside.
Allose is the C3 epimer of glucose, and ribose is the C2 epimer of arabinose.
Which of the following are reducing sugars? Comment on the common name sucrose for table sugar.
(c) α-D-allopyranose
(d) ethyl β-D-ribofuranoside