Textbook Question
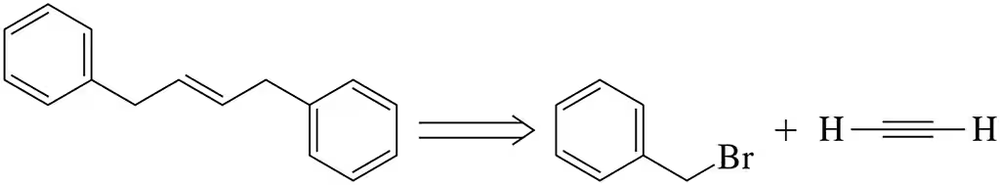
Predict the product of the following reactions.
(a)
944
views

 Verified step by step guidance
Verified step by step guidance


Predict the product of the following reactions.
(a)
Rationalize the difference in Kₑq for the following reactions. Be sure to account for both ∆S and ∆H.
1. Analyze ∆S and ∆H differences." width="737">
Which of the following SN2 and E2 reactions, respectively, is faster? Justify your choice.
(a)