Textbook Question
Explain why an amide ion cannot be used to form a carbanion from an alkane in a reaction that favors products.
1058
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 7 - The Reactions of Alkynes • An Introduction to Multistep Synthesis
Ch. 7 - The Reactions of Alkynes • An Introduction to Multistep Synthesis Problem 23
Problem 23 Verified step by step guidance
Verified step by step guidance

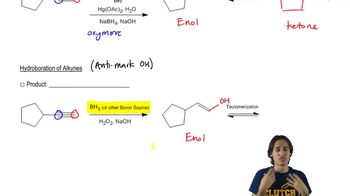

Explain why an amide ion cannot be used to form a carbanion from an alkane in a reaction that favors products.
Which carbocation is more stable?
a.
What are products of the following reactions?
f.
Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products?
Which carbocation is more stable?
b.
How could the following compounds be synthesized from acetylene?
b.