For each of the acid–base reactions in [Section 2.3], compare the pKa values of the acids on either side of the equilibrium arrows to prove that the equilibrium lies in the direction indicated.
1. <IMAGE>
2. <IMAGE>

 Bruice 8th Edition
Bruice 8th Edition Ch. 2 - Acids and Bases: Central to Understanding Organic Chemistry
Ch. 2 - Acids and Bases: Central to Understanding Organic Chemistry Problem 15
Problem 15 Verified step by step guidance
Verified step by step guidance


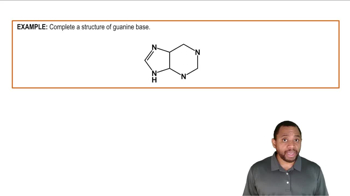
For each of the acid–base reactions in [Section 2.3], compare the pKa values of the acids on either side of the equilibrium arrows to prove that the equilibrium lies in the direction indicated.
1. <IMAGE>
2. <IMAGE>
Draw the conjugate acid of each of the following:
a. CH3CH2OH
b. CH3CH2O−
c.
For each of the acid–base reactions in [Section 2.3], compare the pKa values of the acids on either side of the equilibrium arrows to prove that the equilibrium lies in the direction indicated.
3. <IMAGE>
4. <IMAGE>
Ethyne (HC≡CH) has a pKa value of 25, water has a pKa value of 15.7, and ammonia (NH3) has a pKa value of 36. Draw the equation, showing equilibrium arrows that indicate whether reactants or products are favored, for the acid–base reaction of ethyne with
a. HO-.
a. Which is a stronger base: CH3COO− or HCOO−? (The pKa of CH3COOH is 4.8; the pKa of HCOOH is 3.8.)
b. Which is a stronger base: HO− or -NH2? (The pKa of H2O is 15.7; the pKa of NH3 is 36.)
c. Which is a stronger base: H2O or CH3OH? (The pKa of H3O+ is −1.7; the pKa of CH3O+H2 is −2.5.)
Estimate the pKa values of the following compounds:
c. CH3CH2COOH
d. CH3CH2CH2N+H3