Textbook Question
For each of the following molecules, draw a 3-D representation.
(c) CHBr3
1211
views


 Verified step by step guidance
Verified step by step guidance
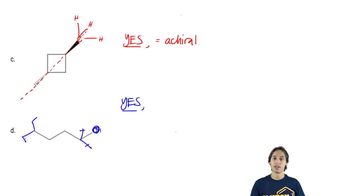


For each of the following molecules, draw a 3-D representation.
(c) CHBr3
For each of the following molecules, draw a 3-D representation.
(d) CHClBrI
Identify all planes of symmetry in the following molecules/conformations, if any.
(a)
Identify all planes of symmetry in the following molecules/conformations, if any.
(c)
Identify all planes of symmetry in the following molecules/conformations, if any.
(f)
In Chapters 8 and 17 we learn two reactions for the synthesis of the alcohol shown. (a) Show a mechanism for each of the reactions. (b) If you were designing a synthetic route, which would be considered more sustainable? Consider all factors. [Assume the starting organic molecules are equally green.]