Textbook Question
Which stereoisomers are optically inactive?
2048
views

 Verified step by step guidance
Verified step by step guidance
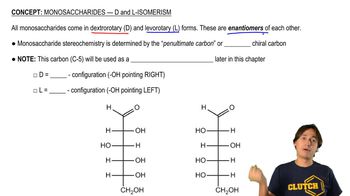


Which stereoisomers are optically inactive?
Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
a.
b.
What is the configuration of the asymmetric centers in the following compounds?
a.
b.
Identify
5. achiral compounds
6. meso compounds
7. enantiomers
8. diastereomers
Identify
1. constitutional isomers
2. stereoisomers
3. cis–trans isomers
4. chiral compounds
A compound has a specific rotation of -39.0. A solution of the compound (0.187 g/100 mL) has an observed rotation of -6.52° when placed in a polarimeter tube 10 cm long. What is the percent of each enantiomer in the solution?