Show the products of the following acetylide alkylation reactions. [Make sure your product has the correct number of carbons.]
(c)

 Mullins 1st Edition
Mullins 1st Edition Ch. 10 - Alkynes: Electrophilic Addition and Redox Reactions
Ch. 10 - Alkynes: Electrophilic Addition and Redox Reactions Problem 47a
Problem 47a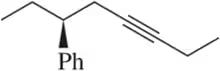
 Verified step by step guidance
Verified step by step guidance



Show the products of the following acetylide alkylation reactions. [Make sure your product has the correct number of carbons.]
(c)
Suggest a method for synthesizing the following alkynes using an alkyne and an alkyl halide. [There are two correct answers for each product.]
(c)
Show the products of the following acetylide alkylation reactions. [Make sure your product has the correct number of carbons.]
(d)
Beginning with the molecules on the left, provide a synthesis of the molecule on the right. The ideal number of steps is indicated over the reaction arrow, although there may be alternate routes worth considering.
(b)
Complete the following synthesis by providing the necessary reagents.
Beginning with the molecules on the left, provide a synthesis of the molecule on the right. The ideal number of steps is indicated over the reaction arrow, although there may be alternate routes worth considering.
(a)