Textbook Question
Draw the structure for each of the following:
h. cyclohexanecarbonyl chloride
594
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 15 - Reactions of Carboxylic Acids and Carboxylic Acid Derivatives
Ch. 15 - Reactions of Carboxylic Acids and Carboxylic Acid Derivatives Problem 5
Problem 5 Verified step by step guidance
Verified step by step guidance
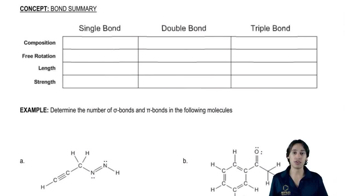
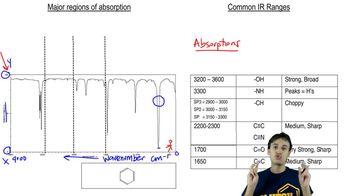
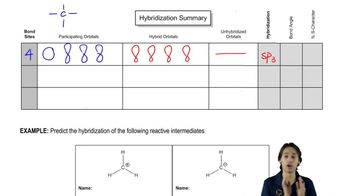
Draw the structure for each of the following:
h. cyclohexanecarbonyl chloride
What reagent should be used to carry out the following reaction?
b. What is the product of the reaction of acetamide with HO−? The pKa of NH3 is 36; the pKa of H2O is 15.7.
Which is longer, the carbon–oxygen single bond in a carboxylic acid or the carbon–oxygen bond in an alcohol? Why?
Draw the structure for each of the following:
i. α-chlorovaleric acid
Which is a correct statement?
A. The delocalization energy of an ester is about 18 kcal/mol, and the delocalization energy of an amide is about 10 kcal/mol.
B. The delocalization energy of an ester is about 10 kcal/mol, and the delocalization energy of an amide is about 18 kcal/mol.