The first step of a reaction called electrophilic aromatic substitution is as follows:
If this step is rate-determining for the overall reaction, which benzene derivative would you expect to react most quickly? Which would react most slowly?
(a)

 Mullins 1st Edition
Mullins 1st Edition Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry
Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry Problem 59
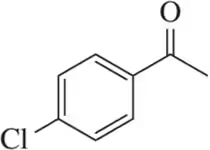
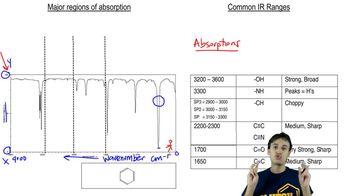
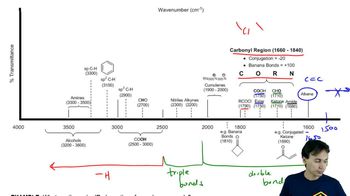
Problem 59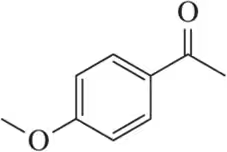


 Verified step by step guidance
Verified step by step guidance

The first step of a reaction called electrophilic aromatic substitution is as follows:
If this step is rate-determining for the overall reaction, which benzene derivative would you expect to react most quickly? Which would react most slowly?
(a)
For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(d)
Justify the carbonyl stretching frequencies indicated for 3-chlorobenzaldehyde and 3-fluorobenzaldehyde.
Justify the carbonyl stretching frequencies indicated for benzaldehyde and 4-methoxybenzaldehyde.
For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(b)
Justify the carbonyl stretching frequencies for a series of methoxybenzaldehydes. Specifically, why are the 2- and 4-methoxy derivatives similar to each other but different from the 3-methoxy derivative?