Textbook Question
Choose the bond in each pair that you expect to vibrate at the higher wavenumber.
(a) C―N vs. C = N
1009
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry
Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry Problem 15b
Problem 15b Verified step by step guidance
Verified step by step guidance
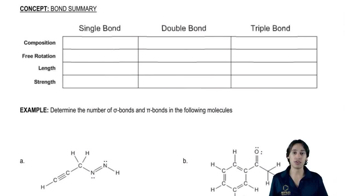


Choose the bond in each pair that you expect to vibrate at the higher wavenumber.
(a) C―N vs. C = N
Given the IR spectrum, suggest what functional groups might correspond to the given molecular formula.
(b) C3H7NO
<IMAGE>
Would you expect an acetylenic C―H to absorb at a higher or lower wavenumber than the C―H in ethene?
Calculate the reduced mass for the following bonds.
(a) C―H
Calculate the reduced mass for the following bonds.
(c) C―Cl
Given the IR spectrum, suggest what functional groups might correspond to the given molecular formula.
(d) C₇H₅O
<IMAGE>