Textbook Question
For each compound,
(1) classify the nitrogen-containing functional groups.
(2) provide an acceptable name.
(a)
(b)
(c)
1020
views

 Verified step by step guidance
Verified step by step guidance

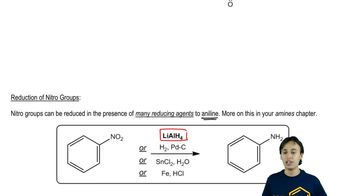

For each compound,
(1) classify the nitrogen-containing functional groups.
(2) provide an acceptable name.
(a)
(b)
(c)
For each compound,
(1) classify the nitrogen-containing functional groups.
(2) provide an acceptable name.
(g)
(h)
Show how you would accomplish the following synthetic conversions.
(d) pentanoic acid → hexan-1-amine
Show how you would accomplish the following synthetic conversions.
(e) (R)-2-bromobutane → (S)-butan-2-amine
Show how you would accomplish the following synthetic conversions.
(f) (R)-2-bromobutane → (S)-2-methylbutan-1-amine
Show how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene and toluene as your aromatic starting materials.
(d) m-aminobenzoic acid